
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
 | |
| Clinical data | |
|---|---|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | Unknown but effects last much longer than other azapirones, up to 23 hours after a single dose in human clinical studies.[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
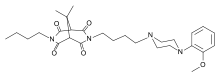
| Formula | C28H40N4O5 |
| Molar mass | 512.651 g·mol−1 |
| 3D model (JSmol) | |
| |
Umespirone (KC-9172) is a drug of the azapirone class which possesses anxiolytic and antipsychotic properties.[2][3][4][5] It behaves as a 5-HT1A receptor partial agonist (Ki = 15 nM), D2 receptor partial agonist (Ki = 23 nM), and α1-adrenoceptor receptor antagonist (Ki = 14 nM), and also has weak affinity for the sigma receptor (Ki = 558 nM).[2][6][7] Unlike many other anxiolytics and antipsychotics, umespirone produces minimal sedation, cognitive/memory impairment, catalepsy, and extrapyramidal symptoms.[1][5][6]
Synthesisedit

The condensation between ethyl cyanoacetate (1) and acetone gives ethylisopropylidenecyanoacetate 759-58-0 (2). This product is reacted with N-butylcyanoacetamide 39581-21-0 (3) in sodium methoxide solution to give N-butyl-2,4-dicyano-3,3-dimethylglutarimide, CID:10681941 (4). The glutarimide is cyclized with phosphoric acid to yield 3-butyl-9,9-dimethyl-3,7-diazabicyclo3.3.1nonane-2,4,6,8-tetraone, https://pubchem.ncbi.nlm.nih.gov/compound/10825633 CID:10825633 (5).
The reaction between 1-(o-anisyl)piperazine 35386-24-4 (6) and 1,4-dibromobutane 110-52-1 (7) gives the Quat salt CID:15895413(8).
Convergent synthesis (in the presence of potassium carbonate) affords Umespirone (KC-9172) (9).
See alsoedit
Referencesedit
- ^ a b Holland RL, Wesnes K, Dietrich B (1994). "Single dose human pharmacology of umespirone". European Journal of Clinical Pharmacology. 46 (5): 461–8. doi:10.1007/bf00191912. PMID 7957544. S2CID 12117650.
- ^ a b Barnes NM, Costall B, Domeney AM, et al. (September 1991). "The effects of umespirone as a potential anxiolytic and antipsychotic agent". Pharmacology Biochemistry and Behavior. 40 (1): 89–96. doi:10.1016/0091-3057(91)90326-W. PMID 1685786. S2CID 9762359.
- ^ Ruhland M, Krähling H, Fuchs A, Schön U (November 1988). "KC 9172 (free base of KC 7218)--an antipsychotic/anxiolytic compound. I. Antipsychotic and anxiolytic activity in comparison with chlorpromazine, clozapine, diazepam and buspirone". Pharmacopsychiatry. 21 (6): 396–8. doi:10.1055/s-2007-1017024. PMID 2907649. S2CID 260241523.
- ^ Krähling H, Fuchs A, Ruhland M, Schön U, Mol F, Tulp M (November 1988). "KC 9172 (free base of KC 7218)--an antipsychotic/anxiolytic compound. II. Discrimination from typical neuroleptics and benzodiazepine-like minor tranquilizers". Pharmacopsychiatry. 21 (6): 399–401. doi:10.1055/s-2007-1017025. PMID 2907650. S2CID 260241655.
- ^ a b Schmidt WJ, Krähling H, Ruhland M (1991). "Antagonism of AP-5-induced sniffing stereotypy links umespirone to atypical antipsychotics". Life Sciences. 48 (6): 499–505. doi:10.1016/0024-3205(91)90464-M. PMID 1671523.
- ^ a b Ahlenius S, Wijkström A (November 1992). "Mixed agonist-antagonist properties of umespirone at neostriatal dopamine receptors in relation to its behavioral effects in the rat". European Journal of Pharmacology. 222 (1): 69–74. doi:10.1016/0014-2999(92)90464-F. PMID 1361441.
- ^ Itzhak Y, Ruhland M, Krähling H (February 1990). "Binding of umespirone to the sigma receptor: evidence for multiple affinity states". Neuropharmacology. 29 (2): 181–4. doi:10.1016/0028-3908(90)90058-Y. PMID 1970425. S2CID 54326248.
- ^ Kr鋒ling, H.; Krijzer, F. Drugs Fut 1991,16(5),437.
- ^ DE3529872 idem Uwe Schoen, Wolfgang Kehrbach, Werner Benson, Andreas Fuchs, Michael Ruhland, U.S. patent 4,771,044 (1988 to Kali-Chemie Pharma Gmbh).
