
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Clear Eyes, Cleari, Naphcon-A, Rohto |
| AHFS/Drugs.com | Monograph |
| Routes of administration | Ophthalmic drug administration, nasal administration |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.011.492 |
| Chemical and physical data | |
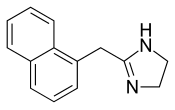
| Formula | C14H14N2 |
| Molar mass | 210.280 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Naphazoline is a medicine used as a decongestant, and a vasoconstrictor added to eye drops to relieve red eye. It has a rapid action in reducing swelling when applied to mucous membranes. It is a sympathomimetic agent with marked alpha adrenergic activity that acts on alpha-receptors in the arterioles of the conjunctiva to produce constriction, resulting in decreased congestion.
It was patented in 1934 and came into medical use in 1942.[1]
Medical uses

Nasal administration
Nasal decongestant.[2]
Ophthalmic drug administration
Eye drops (brand names Clear Eyes, and Cleari) narrowing swollen blood vessels (ophthalmic arteries, and ophthalmic veins) to relieve red eye.[2]
Temporary red eye can safely be treated when the cause of the redness is established (e.g. cannabis induced corneal vasodilation).[3] However, continuous use is not recommended without knowing an underlying condition.
Side effects
A few warnings and contraindications that apply to all naphazoline-containing substances intended for medicinal use are:
- Hypersensitivity to naphazoline
- Use in infants and children can result in central nervous system depression, leading to coma and marked reduction in body temperature
- Should be used with caution in patients with severe cardiovascular disease including cardiac arrhythmia and in patients with diabetes, especially those with a tendency toward diabetic ketoacidosis
- A possible association with stroke has been suggested.[4]
Nasal administration
- Extended use may cause rhinitis medicamentosa, a condition of rebound nasal congestion.
Ophthalmic drug administration
- Stinging
- Discomfort
- Irritation
- Increased red eyes
- Blurred vision
- Mydriasis
- Punctate keratitis
- Lacrimation (tears)
- Increased intraocular pressure
Contraindications
- Patients taking MAO inhibitors can experience a severe hypertensive crisis if given a sympathomimetic drug such as naphazoline HCl
- Drug interactions can occur with anaesthetics that sensitize the myocardium to sympathomimetics (e.g. cyclopropane or halothane cautiously)
- Exercise caution when applying prior to use of phenylephrine.
Pharmacology
Naphazoline is a mixed α1- and α2-adrenergic receptor agonist.[2]
Chemistry
The non-hydrochloride form of Naphazoline has the molecular formula C14H14N2 and a molar mass of 210.28 g/mol. The HCl salt form has a molar mass of 246.73 g/mol.
Society and culture
It is an active ingredient in several over-the-counter eye drop formulations including Clear Eyes, Rohto, Eucool, and Naphcon-A.[7]
Illicit use
The nasal or ophthalmic form of naphazoline has been abused by heroin or cocaine drug addicts.[8][9] It's used as CNS stimulant and vasoconstrictor to enhance primary drug effects.[9]
References
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 552. ISBN 9783527607495.
- ^ a b c Hosten LO, Snyder C (2020). "Over-the-Counter Ocular Decongestants in the United States - Mechanisms of Action and Clinical Utility for Management of Ocular Redness". Clinical Optometry. 12: 95–105. doi:10.2147/OPTO.S259398. PMC 7399465. PMID 32801982.
- ^ Yazulla S (September 2008). "Endocannabinoids in the retina: from marijuana to neuroprotection". Progress in Retinal and Eye Research. 27 (5): 501–526. doi:10.1016/j.preteyeres.2008.07.002. PMC 2584875. PMID 18725316.
- ^ Zavala JA, Pereira ER, Zétola VH, Teive HA, Nóvak EM, Werneck LC (September 2004). "Hemorrhagic stroke after naphazoline exposition: case report". Arquivos de Neuro-Psiquiatria. 62 (3B): 889–891. doi:10.1590/S0004-282X2004000500030. PMID 15476091.
- ^ "Naphazoline - FDA prescribing information, side effects and uses". Drugs.com.
- ^ "naphazoline ophthalmic (eye): Uses, Side Effects, Interactions, Pictures, Warnings & Dosing - WebMD". www.webmd.com.
- ^ Green SM (2008). "Ophthalmology: Naphazoline". Tarascon Pocket Pharmacopoeia 2009. Jones and Bartlett. ISBN 978-0-7637-6572-9.
- ^ van Montfrans GA, van Steenwijk RP, Vyth A, Borst C (1981). "Intravenous naphazoline intoxication". Acta Medica Scandinavica. 209 (5): 429–430. doi:10.1111/j.0954-6820.1981.tb11622.x. PMID 7246278.
- ^ a b "Naphazoline abuse". Reactions Weekly. 1815 (1): 251. 2020-08-01. doi:10.1007/s40278-020-81577-1. ISSN 1179-2051. S2CID 195174995.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
