
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
This article needs additional citations for verification. (September 2014) |
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Wytensin |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a686003 |
| ATC code |
|
| Pharmacokinetic data | |
| Protein binding | 90% |
| Elimination half-life | 6 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.023.410 |
| Chemical and physical data | |
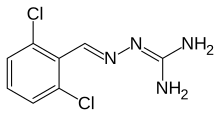
| Formula | C8H8Cl2N4 |
| Molar mass | 231.08 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Guanabenz (pronounced GWAHN-a-benz, sold under the trade name Wytensin) is an alpha agonist that is selective to the alpha-2 adrenergic receptor. Guanabenz is used as an antihypertensive drug used in the treatment of high blood pressure (hypertension).[1][2]
The most common side effects during guanabenz therapy are dizziness, drowsiness, dry mouth, headache and weakness.[3]
Guanabenz can make one drowsy or less alert, therefore driving or operating dangerous machinery is not recommended.
Research
Guanabenz also has some anti-inflammatory properties in different pathological situations, including multiple sclerosis.[4]
Guanabenz was found in one study to exert an inhibitory effect by decreasing the abundance of the enzyme CH25H, a cholesterol hydroxylase linked to antiviral immunity. Therefore, it is suggested that the drug and similar compounds could be used to treat type I interferon-dependent pathologies and that the CH25H enzyme could be a therapeutic target to control these diseases,[5] including amyotrophic lateral sclerosis.
See also
References
- ^ Walker BR, Hare LE, Deitch MW (1982). "Comparative antihypertensive effects of guanabenz and clonidine". The Journal of International Medical Research. 10 (1): 6–14. doi:10.1177/030006058201000102. PMID 7037502. S2CID 2139809.[permanent dead link]
- ^ Bonham AC, Trapani AJ, Portis LR, Brody MJ (December 1984). "Studies on the mechanism of the central antihypertensive effect of guanabenz and clonidine". Journal of Hypertension Supplement. 2 (3): S543–S546. PMID 6599714.[permanent dead link]
- ^ "Guanabenz | The Merck Index Online". www.rsc.org. Retrieved 2023-04-17.
- ^ Way SW, Podojil JR, Clayton BL, Zaremba A, Collins TL, Kunjamma RB, et al. (March 2015). "Pharmaceutical integrated stress response enhancement protects oligodendrocytes and provides a potential multiple sclerosis therapeutic". Nature Communications. 6: 6532. Bibcode:2015NatCo...6.6532W. doi:10.1038/ncomms7532. PMC 4360920. PMID 25766071.
- ^ Perego J, Mendes A, Bourbon C, Camosseto V, Combes A, Liu H, et al. (January 2018). "Guanabenz inhibits TLR9 signaling through a pathway that is independent of eIF2α dephosphorylation by the GADD34/PP1c complex". Science Signaling. 11 (514): eaam8104. doi:10.1126/scisignal.aam8104. hdl:10773/27856. PMID 29363586. S2CID 13680678.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
