A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |||
| Identifiers | |||
|---|---|---|---|
3D model (JSmol)
|
|||
| |||
| |||
| Properties | |||
| Br3Np | |||
| Molar mass | 477 g·mol−1 | ||
| Appearance | green solid[1] | ||
| Density | 6.62 g·cm−3[2] | ||
| Structure | |||
| α-NpBr3: hexagonal β-NpBr3: orthorhombic | |||
| α-NpBr3: P63/m (No. 176) β-NpBr3: Ccmm (No. 63) | |||
a = 791.7 pm (α), 411 pm (β), b = 791.7 pm (α), 1265 pm (β), c = 438.2 pm (α), 915 pm (β)
| |||
| Related compounds | |||
Other anions
|
neptunium(III) fluoride neptunium(III) chloride neptunium(III) iodide | ||
Other cations
|
uranium(III) bromide plutonium(III) bromide | ||
Related compounds
|
neptunium(IV) bromide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Neptunium(III) bromide is a bromide of neptunium, with the chemical formula of NpBr3.
Preparation
Neptunium(III) bromide can be prepared by reacting neptunium dioxide and aluminium bromide:[3]
- 6 NpO2 + 8 AlBr3 → 6 NpBr3 + 4 Al2O3 + 3 Br2
Properties
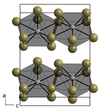
Neptunium(III) bromide is a green solid. It can crystallize in two crystal systems:
- α-NpBr3 is hexagonal with lattice parameters a = 791.7 pm and c = 438.2 pm.[2] It has the same structure as uranium trichloride.
- β-NpBr3 is orthorhombic with lattice parameters a = 411 pm, b = 1265 pm and c = 915 pm.[2] It has the same structure as the bromides from plutonium to californium.
Neptunium(III) bromide also has a green hexahydrate, which is monoclinic.[3]
Reactions
At 425 °C, neptunium(III) bromide bromide can be further brominated by bromine to form neptunium(IV) bromide.[2]
- 2 NpBr3 + Br2 → 2 NpBr4
References
- ^ Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, p. 1969, ISBN 0-12-352651-5
- ^ a b c d Gmelins Handbuch der anorganischen Chemie, System No. 71, Transurane, Teil C, S. 148–150.
- ^ a b Georg Brauer (Hrsg.), unter Mitarbeit von Marianne Baudler u. a.: Handbuch der Präparativen Anorganischen Chemie. 3., umgearbeitete Auflage. Band II, Ferdinand Enke, Stuttgart 1978, ISBN 3-432-87813-3, S. 1268.
External reading
- Yoshida, Zenko; Johnson, Stephen G.; Kimura, Takaumi; Krsul, John R. (2006), Morss, Lester R.; Edelstein, Norman M.; Fuger, Jean (eds.), "Neptunium", The Chemistry of the Actinide and Transactinide Elements, Dordrecht: Springer Netherlands, pp. 699–812, doi:10.1007/1-4020-3598-5_6, ISBN 978-1-4020-3555-5, retrieved 2023-12-23
- Keller, C. (1969), "Die Chemie des Neptuniums", Anorganische Chemie, Fortschritte der Chemischen Forschung (in German), vol. 13/1, Berlin/Heidelberg: Springer-Verlag, pp. 1–124, doi:10.1007/bfb0051170, ISBN 978-3-540-04488-8, retrieved 2023-12-23
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk