
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |

| |
| Names | |
|---|---|
| IUPAC name
Cesium bromide
| |
| Other names
Cesium bromide,
Caesium(I) bromide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.209 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CsBr | |
| Molar mass | 212.809 g/mol[1] |
| Appearance | White solid |
| Density | 4.43 g/cm3[1] |
| Melting point | 636 °C (1,177 °F; 909 K)[1] |
| Boiling point | 1,300 °C (2,370 °F; 1,570 K)[1] |
| 1230 g/L (25 °C)[1] Disputed.
420 g/L (11 °C) See References | |
| -67.2·10−6 cm3/mol[2] | |
Refractive index (nD)
|
1.8047 (0.3 μm) 1.6974 (0.59 μm) 1.6861 (0.75 μm) 1.6784 (1 μm) 1.6678 (5 μm) 1.6439 (20 μm)[3] |
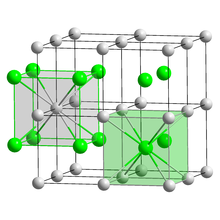
| Structure | |
| CsCl, cP2 | |
| Pm3m, No. 221[4] | |
a = 0.4291 nm
| |
Lattice volume (V)
|
0.0790 nm3 |
Formula units (Z)
|
1 |
| Cubic (Cs+) Cubic (Br−) | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H315, H319, H335 | |
| P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
1400 mg/kg (oral, rat)[5] |
| Related compounds | |
Other anions
|
Caesium fluoride Caesium chloride Caesium iodide Caesium astatide |
Other cations
|
Sodium bromide Potassium bromide Rubidium bromide Francium bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Caesium bromide or cesium bromide is an ionic compound of caesium and bromine with the chemical formula CsBr. It is a white or transparent solid with melting point at 636 °C that readily dissolves in water. Its bulk crystals have the cubic CsCl structure, but the structure changes to the rocksalt type in nanometer-thin film grown on mica, LiF, KBr or NaCl substrates.[6]
Synthesis
Caesium bromide can be prepared via following reactions:
- CsOH (aq) + HBr (aq) → CsBr (aq) + H2O (l)
- Cs2(CO3) (aq) + 2 HBr (aq) → 2 CsBr (aq) + H2O (l) + CO2 (g)
- Direct synthesis:
- 2 Cs (s) + Br2 (g) → 2 CsBr (s)
The direct synthesis is a vigorous reaction of caesium with bromine. Due to its high cost, it is not used for preparation.
Uses
Caesium bromide is sometimes used in optics as a beamsplitter component in wide-band spectrophotometers.
References
- ^ a b c d e Haynes, p. 4.57
- ^ Haynes, p. 4.132
- ^ Haynes, p. 10.240
- ^ Vallin, J.; Beckman, O.; Salama, K. (1964). "Elastic Constants of CsBr and CsI from 4.2K to Room Temperature". Journal of Applied Physics. 35 (4): 1222. Bibcode:1964JAP....35.1222V. doi:10.1063/1.1713597.
- ^ Caesium bromide. nlm.nih.gov
- ^ Schulz, L. G. (1951). "Polymorphism of cesium and thallium halides". Acta Crystallographica. 4 (6): 487–489. doi:10.1107/S0365110X51001641.
* Crystran Ltd experimental data July 2021 Archived 2012-12-18 at the Wayback Machine
Cited sources
- Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. ISBN 1-4398-5511-0.
External links
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk

