
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9

| |
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.246 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| |
| Molar mass | 184.113 g/mol (anhydrous) 292.204 g/mol (hexahydrate) |
| Appearance | white hygroscopic hexagonal crystals (anhydrous) colorless monoclinic crystals (hexahydrate) |
| Density | 3.72 g/cm3 (anhydrous) 2.07 g/cm3 (hexahydrate) |
| Melting point | 711 °C (1,312 °F; 984 K) 172.4 °C, decomposes (hexahydrate) |
| Boiling point | 1,250 °C (2,280 °F; 1,520 K) |
| 102 g/(100 mL) (anhydrous) 316 g/(100 mL) (0 °C, hexahydrate) | |
| Solubility | ethanol: 6.9 g/(100 mL) methanol: 21.8 g/(100 mL) |
| −72.0·10−6 cm3/mol | |
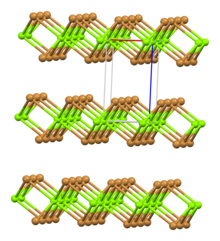
| Structure | |
| Rhombohedral, hP3 | |
| P-3m1, No. 164 | |
| octahedral | |
| Thermochemistry | |
Heat capacity (C)
|
70 J/(mol·K) |
Std molar
entropy (S⦵298) |
117.2 J·mol−1·K−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−524.3 kJ·mol−1 |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | External SDS |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Magnesium bromide is a chemical compound of magnesium and bromine, with the chemical formula MgBr2. It is white and deliquescent crystalline solid. It is often used as a mild sedative and as an anticonvulsant for treatment of nervous disorders.[2] It is water-soluble and somewhat soluble in alcohol. It can be found naturally in small amounts in some minerals such as: bischofite and carnallite, and in sea water, such as that of the Dead Sea.[3][4]
Synthesis
Magnesium bromide can be synthesized by treating with magnesium oxide (and related basic salts) with hydrobromic acid.[4] It can also be made by reacting magnesium carbonate and hydrobromic acids, and collecting the solid left after evaporation.[3]
As suggested by its easy conversion to various hydrates, anhydrous MgBr2 is a Lewis acid. In the coordination polymer with the formula MgBr2(dioxane)2, Mg2+ adopts an octahedral geometry.[5]
Uses
Magnesium bromide is used as a Lewis acid catalyst in some organic synthesis, e.g., in aldol reaction.[6] In organosilicon chemistry, magnesium bromide forms adducts R2SiXMgBr2.[7]
Magnesium bromide also has been used as a tranquilizer.[3]
Magnesium bromide modifies the catalytic properties of palladium on charcoal.[8]
Magnesium bromide hexahydrate has properties as a flame retardant. It was found that if 0.125 mol/L of magnesium bromide hexahydrate was added to a cotton material it acted as a flame retardant.[9]
References
- ^ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. 4–67. ISBN 0-8493-0594-2.
- ^ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- ^ a b c Gruyter, W. Concise Encyclopedia Chemistry, Walter de Gruyter & Company: Berlin, 1993; 612
- ^ a b Lewis, R.J. Hawley’s Condensed Chemical Dictionary, 15th ed.; John Wiley &Sons Inc.:New York, 2007; 777
- ^ Fischer, Reinald; Görls, Helmar; Meisinger, Philippe R.; Suxdorf, Regina; Westerhausen, Matthias (2019). "Structure–Solubility Relationship of 1,4‐Dioxane Complexes of Di(hydrocarbyl)magnesium". Chemistry – A European Journal. 25 (55): 12830–12841. doi:10.1002/chem.201903120. PMC 7027550. PMID 31328293.
- ^ Evans, David A.; Tedrow, Jason S.; Shaw, Jared T.; Downey, C. Wade (2002). "Diastereoselective Magnesium Halide-Catalyzed anti-Aldol Reactions of Chiral N-Acyloxazolidinones". Journal of the American Chemical Society. 124 (3): 392–393. doi:10.1021/ja0119548. PMID 11792206.
- ^ Lim, Young Mook; Cho, Hyeon Mo; Lee, Myong Euy; Baeck, Kyoung Koo (2006). "A Stable Magnesium Bromosilylenoid: Transmetalation of a Lithium Bromosilylenoid by Magnesium Bromide". Organometallics. 25 (21): 4960. doi:10.1021/om060589w.
- ^ Bouzide, Abderrahim (2002). "Magnesium Bromide Mediated Highly Diastereoselective Heterogeneous Hydrogenation of Olefins". Organic Letters. 4 (8): 1347–50. doi:10.1021/ol020032m. PMID 11950359.
- ^ Mostashari, S. M.; Fayyaz, F. (2008). "XRD characterization of the ashes from a burned cellulosic fabric impregnated with magnesium bromide hexahydrate as flame-retardant". Journal of Thermal Analysis and Calorimetry. 92 (3): 845. doi:10.1007/s10973-007-8928-4. S2CID 94416902.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk

