
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Identifiers | |
|---|---|
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.933 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Appearance | colourless solid (anhydrous)[1] white solid (hexahydrate)[2] |
| Density | 5.8 g·cm−3[3] |
| Melting point | 881 °C (1,154 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Dysprosium(III) bromide is an inorganic compound of bromine and dysprosium, with the chemical formula of DyBr3.
Preparation
Dysprosium(III) bromide can be obtained by reacting dysprosium with bromine:[4]
- 2Dy + 3Br2 → 2DyBr3
Dysprosium bromide hexahydrate can be obtained by crystallization from its solution,[2] which can be heated with ammonium bromide in vacuum to obtain the anhydrous form.[1]
Dysprosium(III) oxide and aluminium bromide (in the form of Al2Br6 at a high temperature react a DyAl3Br12, which decomposes to dysprosium(III) bromide at a lower temperature:[5]
- Dy2O3 + Al2Br6 → Al2O3 + 2 DyBr3
Properties
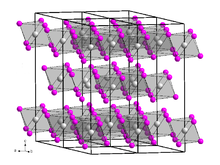
Dysprosium(III) bromide is a white-gray hygroscopic solid that is soluble in water.[6] It has a trigonal crystal structure of the bismuth(III) iodide type with space group R3 (No. 148).[7]
References
- ^ a b c Jantsch, G.; Jawurek, H.; Skalla, N.; Gawalowski, H. Halides of the rare earths. VI. Halides of the terbium and erbium earth groups. Zeitschrift fuer Anorganische und Allgemeine Chemie, 1932. 207. 353-367. ISSN 0044-2313.
- ^ a b D. Brown, S. Fletcher, D. G. Holah (1968). "The preparation and crystallographic properties of certain lanthanide and actinide tribromides and tribromide hexahydrates". Journal of the Chemical Society A: Inorganic, Physical, Theoretical: 1889–1894. doi:10.1039/j19680001889. ISSN 0022-4944. Retrieved 2020-05-29.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Roger Blachnik (Hrsg.): Taschenbuch für Chemiker und Physiker. Band III: Elemente, anorganische Verbindungen und Materialien, Minerale. begründet von Jean d’Ans, Ellen Lax. 4., neubearbeitete und revidierte Auflage. Springer, Berlin 1998, ISBN 3-540-60035-3, S. 442, 1386
- ^ WebElements: Chemical reactions of Dysprosium
- ^ 杨冬梅, 于锦, 蒋军辉,等. 化学气相传输法制备无水溴化镝. 石油化工高等学校学报, 2003, 16(4). doi: 10.3969/j.issn.1006-396X.2003.04.004.
- ^ Dysprosium(III) bromide, ultra dry, 99.99% (REO) at AlfaAesar, accessed on 2013-10-30 (PDF) (JavaScript required).[dead link]
- ^ Ans, Jean d'; Lax, Ellen (December 1997). Taschenbuch für Chemiker und Physiker. Springer. pp. 442, 1386. ISBN 3540600353.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
