
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
The Algar–Flynn–Oyamada reaction is a chemical reaction whereby a chalcone undergoes an oxidative cyclization to form a flavonol.[1][2]
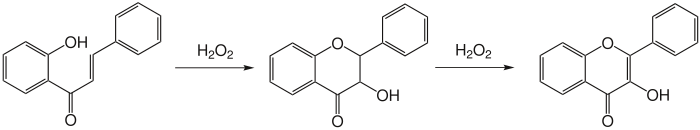
Reaction mechanism
There are several possible mechanisms to explain this reaction; however, these reaction mechanisms have not been elucidated. What is known is that a two-stage mechanism exists. First, dihydroflavonol is formed, which then subsequently oxidizes to form a flavonol.
Proposed mechanisms involving epoxidation of the alkene have been disproven.[3]
The probable mechanisms are thus two possibilities:
- The phenoxide attacks the enone at the beta position, and the alkene directly attacks hydrogen peroxide from the alpha position, forming the dihydroflavonol.
- The phenoxide attacks the enone at the beta position, closing the six-membered ring and forming an enolate intermediate. The enolate then attacks hydrogen peroxide, forming the dihydroflavonol.

See also
References
- ^ Algar, J.; Flynn, J. P. (1934). Proceedings of the Royal Irish Academy. 42B: 1.
{{cite journal}}: CS1 maint: untitled periodical (link) - ^ Oyamada, B. (1935). "A New General Method for the Synthesis of the Derivatives of Flavonol". Bulletin of the Chemical Society of Japan. 10 (5): 182–186. doi:10.1246/bcsj.10.182.
- ^ Gormley, T.R.; O'Sullivan, W.I. (1973). "Flavanoid epoxides—XIII". Tetrahedron. 29 (2): 369–373. doi:10.1016/S0040-4020(01)93304-6. hdl:10197/6996. ISSN 0040-4020.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
