
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
A solvent (from the Latin solvō, "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for polar molecules, and the most common solvent used by living things; all the ions and proteins in a cell are dissolved in water within the cell.
Major uses of solvents are in paints, paint removers, inks, and dry cleaning.[2] Specific uses for organic solvents are in dry cleaning (e.g. tetrachloroethylene); as paint thinners (toluene, turpentine); as nail polish removers and solvents of glue (acetone, methyl acetate, ethyl acetate); in spot removers (hexane, petrol ether); in detergents (citrus terpenes); and in perfumes (ethanol). Solvents find various applications in chemical, pharmaceutical, oil, and gas industries, including in chemical syntheses and purification processes.
Solutions and solvation
When one substance is dissolved into another, a solution is formed.[3] This is opposed to the situation when the compounds are insoluble like sand in water. In a solution, all of the ingredients are uniformly distributed at a molecular level and no residue remains. A solvent-solute mixture consists of a single phase with all solute molecules occurring as solvates (solvent-solute complexes), as opposed to separate continuous phases as in suspensions, emulsions and other types of non-solution mixtures. The ability of one compound to be dissolved in another is known as solubility; if this occurs in all proportions, it is called miscible.
In addition to mixing, the substances in a solution interact with each other at the molecular level. When something is dissolved, molecules of the solvent arrange around molecules of the solute. Heat transfer is involved and entropy is increased making the solution more thermodynamically stable than the solute and solvent separately. This arrangement is mediated by the respective chemical properties of the solvent and solute, such as hydrogen bonding, dipole moment and polarizability.[4] Solvation does not cause a chemical reaction or chemical configuration changes in the solute. However, solvation resembles a coordination complex formation reaction, often with considerable energetics (heat of solvation and entropy of solvation) and is thus far from a neutral process.
When one substance dissolves into another, a solution is formed. A solution is a homogeneous mixture consisting of a solute dissolved into a solvent. The solute is the substance that is being dissolved, while the solvent is the dissolving medium. Solutions can be formed with many different types and forms of solutes and solvents.
Solvent classifications
Solvents can be broadly classified into two categories: polar and non-polar. A special case is elemental mercury, whose solutions are known as amalgams; also, other metal solutions exist which are liquid at room temperature.
Generally, the dielectric constant of the solvent provides a rough measure of a solvent's polarity. The strong polarity of water is indicated by its high dielectric constant of 88 (at 0 °C).[5] Solvents with a dielectric constant of less than 15 are generally considered to be nonpolar.[6]
The dielectric constant measures the solvent's tendency to partly cancel the field strength of the electric field of a charged particle immersed in it. This reduction is then compared to the field strength of the charged particle in a vacuum.[6] Heuristically, the dielectric constant of a solvent can be thought of as its ability to reduce the solute's effective internal charge. Generally, the dielectric constant of a solvent is an acceptable predictor of the solvent's ability to dissolve common ionic compounds, such as salts.
Other polarity scales
Dielectric constants are not the only measure of polarity. Because solvents are used by chemists to carry out chemical reactions or observe chemical and biological phenomena, more specific measures of polarity are required. Most of these measures are sensitive to chemical structure.
The Grunwald–Winstein mY scale measures polarity in terms of solvent influence on buildup of positive charge of a solute during a chemical reaction.
Kosower's Z scale measures polarity in terms of the influence of the solvent on UV-absorption maxima of a salt, usually pyridinium iodide or the pyridinium zwitterion.[7]
Donor number and donor acceptor scale measures polarity in terms of how a solvent interacts with specific substances, like a strong Lewis acid or a strong Lewis base.[8]
The Hildebrand parameter is the square root of cohesive energy density. It can be used with nonpolar compounds, but cannot accommodate complex chemistry.
Reichardt's dye, a solvatochromic dye that changes color in response to polarity, gives a scale of ET(30) values. ET is the transition energy between the ground state and the lowest excited state in kcal/mol, and (30) identifies the dye. Another, roughly correlated scale (ET(33)) can be defined with Nile red.
The polarity, dipole moment, polarizability and hydrogen bonding of a solvent determines what type of compounds it is able to dissolve and with what other solvents or liquid compounds it is miscible. Generally, polar solvents dissolve polar compounds best and non-polar solvents dissolve non-polar compounds best; hence "like dissolves like". Strongly polar compounds like sugars (e.g. sucrose) or ionic compounds, like inorganic salts (e.g. table salt) dissolve only in very polar solvents like water, while strongly non-polar compounds like oils or waxes dissolve only in very non-polar organic solvents like hexane. Similarly, water and hexane (or vinegar and vegetable oil) are not miscible with each other and will quickly separate into two layers even after being shaken well.
Polarity can be separated to different contributions. For example, the Kamlet-Taft parameters are dipolarity/polarizability (π*), hydrogen-bonding acidity (α) and hydrogen-bonding basicity (β). These can be calculated from the wavelength shifts of 3–6 different solvatochromic dyes in the solvent, usually including Reichardt's dye, nitroaniline and diethylnitroaniline. Another option, Hansen solubility parameters, separates the cohesive energy density into dispersion, polar, and hydrogen bonding contributions.
Polar protic and polar aprotic
Solvents with a dielectric constant (more accurately, relative static permittivity) greater than 15 (i.e. polar or polarizable) can be further divided into protic and aprotic. Protic solvents, such as water, solvate anions (negatively charged solutes) strongly via hydrogen bonding. Polar aprotic solvents, such as acetone or dichloromethane, tend to have large dipole moments (separation of partial positive and partial negative charges within the same molecule) and solvate positively charged species via their negative dipole.[9] In chemical reactions the use of polar protic solvents favors the SN1 reaction mechanism, while polar aprotic solvents favor the SN2 reaction mechanism. These polar solvents are capable of forming hydrogen bonds with water to dissolve in water whereas non-polar solvents are not capable of strong hydrogen bonds.
Physical properties
Properties table of common solvents
The solvents are grouped into nonpolar, polar aprotic, and polar protic solvents, with each group ordered by increasing polarity. The properties of solvents which exceed those of water are bolded.
| Solvent | Chemical formula | Boiling point[10] (°C) |
Dielectric constant[11] | Density (g/mL) |
Dipole moment (D) |
|---|---|---|---|---|---|
Nonpolar hydrocarbon solvents | |||||
| Pentane | 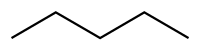
CH3CH2CH2CH2CH3 |
36.1 | 1.84 | 0.626 | 0.00 |
| Hexane | 
CH3CH2CH2CH2CH2CH3 |
69 | 1.88 | 0.655 | 0.00 |
| Benzene | 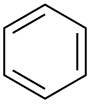 C6H6 |
80.1 | 2.3 | 0.879 | 0.00 |
| Heptane | 
H3C(CH2)5CH3 |
98.38 | 1.92 | 0.680 | 0.0 |
| Toluene | 
C6H5-CH3 |
111 | 2.38 | 0.867 | 0.36 |
Nonpolar ether solvents | |||||
| 1,4-Dioxane |  C4H8O2 |
101.1 | 2.3 | 1.033 | 0.45 |
| Diethyl ether | 
CH3CH2-O-CH2CH3 |
34.6 | 4.3 | 0.713 | 1.15 |
| Tetrahydrofuran (THF) | 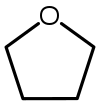 C4H8O |
66 | 7.5 | 0.886 | 1.75 |
Nonpolar chlorocarbon solvents | |||||
| Chloroform | 
CHCl3 |
61.2 | 4.81 | 1.498 | 1.04 |
| Polar aprotic solvents | |||||
| Dichloromethane (DCM) | 
CH2Cl2 |
39.6 | 9.1 | 1.3266 | 1.60 |
| Ethyl acetate |  CH3-C(=O)-O-CH2-CH3 |
77.1 | 6.02 | 0.894 | 1.78 |
| Acetone |  CH3-C(=O)-CH3 |
56.1 | 21 | 0.786 | 2.88 |
| Dimethylformamide (DMF) | 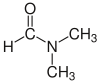 H-C(=O)N(CH3)2 |
153 | 38 | 0.944 | 3.82 |
| Acetonitrile (MeCN) | CH3-C≡N |
82 | 37.5 | 0.786 | 3.92 |
| Dimethyl sulfoxide (DMSO) |  CH3-S(=O)-CH3 |
189 | 46.7 | 1.092 | 3.96 |
| Nitromethane | 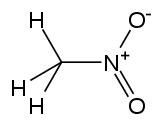
CH3-NO2 |
100–103 | 35.87 | 1.1371 | 3.56 |
| Propylene carbonate | 
C4H6O3 |
240 | 64.0 | 1.205 | 4.9 |
Polar protic solvents | |||||
| Ammonia | 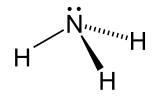
NH3 |
-33.3 | 17 | 0.674
(at -33.3 °C) |
1.42 |
| Formic acid | 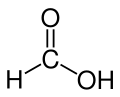 H-C(=O)OH |
100.8 | 58 | 1.21 | 1.41 |
| n-Butanol | CH3CH2CH2CH2OH |
117.7 | 18 | 0.810 | 1.63 |
| Isopropyl alcohol (IPA) | 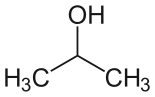 CH3-CH(-OH)-CH3 |
82.6 | 18 | 0.785 | 1.66 |
| n-Propanol | 
CH3CH2CH2OH |
97 | 20 | 0.803 | 1.68 |
| Ethanol | CH3CH2OH |
78.2 | 24.55 | 0.789 | 1.69 |
| Methanol | 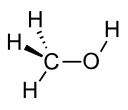
CH3OH |
64.7 | 33 | 0.791 | 1.70 |
| Acetic acid |  CH3-C(=O)OH |
118 | 6.2 | 1.049 | 1.74 |
| Water |  H-O-H |
100 | 80 | 1.000 | 1.85 |
The ACS Green Chemistry Institute maintains a tool for the selection of solvents based on a principal component analysis of solvent properties.[12]
Hansen solubility parameter values
The Hansen solubility parameter (HSP) values[13][14] are based on dispersion bonds (δD), polar bonds (δP) and hydrogen bonds (δH). These contain information about the inter-molecular interactions with other solvents and also with polymers, pigments, nanoparticles, etc. This allows for rational formulations knowing, for example, that there is a good HSP match between a solvent and a polymer. Rational substitutions can also be made for "good" solvents (effective at dissolving the solute) that are "bad" (expensive or hazardous to health or the environment). The following table shows that the intuitions from "non-polar", "polar aprotic" and "polar protic" are put numerically – the "polar" molecules have higher levels of δP and the protic solvents have higher levels of δH. Because numerical values are used, comparisons can be made rationally by comparing numbers. For example, acetonitrile is much more polar than acetone but exhibits slightly less hydrogen bonding.
| Solvent | Chemical formula | δD Dispersion | δP Polar | δH Hydrogen bonding |
|---|---|---|---|---|
Non-polar solvents | ||||
| n-Hexane | CH3CH2CH2CH2CH2CH3 | 14.9 | 0.0 | 0.0 |
| Benzene | C6H6 | 18.4 | 0.0 | 2.0 |
| Toluene | C6H5-CH3 | 18.0 | 1.4 | 2.0 |
| Diethyl ether | CH3CH2-O-CH2CH3 | 14.5 | 2.9 | 4.6 |
| Chloroform | CHCl3 | 17.8 | 3.1 | 5.7 |
| 1,4-Dioxane | /-CH2-CH2-O-CH2-CH2-O-\ | 17.5 | 1.8 | 9.0 |
Polar aprotic solvents | ||||
| Ethyl acetate | CH3-C(=O)-O-CH2-CH3 | 15.8 | 5.3 | 7.2 |
| Tetrahydrofuran (THF) | /-CH2-CH2-O-CH2-CH2-\ | 16.8 | 5.7 | 8.0 |
| Dichloromethane | CH2Cl2 | Zdroj:https://en.wikipedia.org?pojem=Solvent|||
