A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9

| |||
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Water
| |||
| Systematic IUPAC name
Oxidane | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3587155 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.028.902 | ||
| EC Number |
| ||
| 117 | |||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
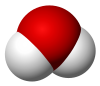
| H 2O | |||
| Molar mass | 18.01528(33) g/mol | ||
| Appearance | Almost colorless or white crystalline solid, almost colorless liquid, with a hint of blue, colorless gas[3] | ||
| Odor | Odorless | ||
| Density | |||
| Melting point | 0.00 °C (32.00 °F; 273.15 K) [b] | ||
| Boiling point | 99.98 °C (211.96 °F; 373.13 K)[16][b] | ||
| Solubility | Poorly soluble in haloalkanes, aliphatic and aromatic hydrocarbons, ethers.[7] Improved solubility in carboxylates, alcohols, ketones, amines. Miscible with methanol, ethanol, propanol, isopropanol, acetone, glycerol, 1,4-dioxane, tetrahydrofuran, sulfolane, acetaldehyde, dimethylformamide, dimethoxyethane, dimethyl sulfoxide, acetonitrile. Partially miscible with diethyl ether, methyl ethyl ketone, dichloromethane, ethyl acetate, bromine. | ||
| Vapor pressure | 3.1690 kilopascals or 0.031276 atm at 25 °C[8] | ||
| Acidity (pKa) | 13.995[9][10][a] | ||
| Basicity (pKb) | 13.995 | ||
| Conjugate acid | Hydronium H3O+ (pKa = 0) | ||
| Conjugate base | Hydroxide OH– (pKb = 0) | ||
| Thermal conductivity | Zdroj:https://en.wikipedia.org?pojem=Water_(molecule)|||