
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9

The atmosphere of Earth is the layer of gases, known collectively as air, retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The atmosphere of Earth creates pressure, absorbs most meteoroids and ultraviolet solar radiation, warms the surface through heat retention (greenhouse effect), and reduces temperature extremes between day and night (the diurnal temperature variation), maintaining conditions allowing life and liquid water to exist on the Earth's surface.
As of 2023, by mole fraction (i.e., by number of molecules), dry air contains 78.08% nitrogen, 20.95% oxygen, 0.93% argon, 0.04% carbon dioxide, and small amounts of other gases.[8] Air also contains a variable amount of water vapor, on average around 1% at sea level, and 0.4% over the entire atmosphere. Air composition, temperature, and atmospheric pressure vary with altitude. Within the atmosphere, air suitable for use in photosynthesis by terrestrial plants and breathing of terrestrial animals is found only in Earth's troposphere.[citation needed]
Earth's early atmosphere consisted of gases in the solar nebula, primarily hydrogen. The atmosphere changed significantly over time, affected by many factors such as volcanism, life, and weathering. Recently, human activity has also contributed to atmospheric changes, such as global warming, ozone depletion and acid deposition.
The atmosphere has a mass of about 5.15×1018 kg,[9] three quarters of which is within about 11 km (6.8 mi; 36,000 ft) of the surface. The atmosphere becomes thinner with increasing altitude, with no definite boundary between the atmosphere and outer space. The Kármán line, at 100 km (62 mi) or 1.57% of Earth's radius, is often used as the border between the atmosphere and outer space. Atmospheric effects become noticeable during atmospheric reentry of spacecraft at an altitude of around 120 km (75 mi). Several layers can be distinguished in the atmosphere, based on characteristics such as temperature and composition.
The study of Earth's atmosphere and its processes is called atmospheric science (aerology), and includes multiple subfields, such as climatology and atmospheric physics. Early pioneers in the field include Léon Teisserenc de Bort and Richard Assmann.[10] The study of historic atmosphere is called paleoclimatology.
Composition

The three major constituents of Earth's atmosphere are nitrogen, oxygen, and argon. Water vapor accounts for roughly 0.25% of the atmosphere by mass. The concentration of water vapor (a greenhouse gas) varies significantly from around 10 ppm by mole fraction in the coldest portions of the atmosphere to as much as 5% by mole fraction in hot, humid air masses, and concentrations of other atmospheric gases are typically quoted in terms of dry air (without water vapor).[11]: 8 The remaining gases are often referred to as trace gases,[12] among which are other greenhouse gases, principally carbon dioxide, methane, nitrous oxide, and ozone. Besides argon, already mentioned, other noble gases, neon, helium, krypton, and xenon are also present. Filtered air includes trace amounts of many other chemical compounds. Many substances of natural origin may be present in locally and seasonally variable small amounts as aerosols in an unfiltered air sample, including dust of mineral and organic composition, pollen and spores, sea spray, and volcanic ash. Various industrial pollutants also may be present as gases or aerosols, such as chlorine (elemental or in compounds), fluorine compounds and elemental mercury vapor. Sulfur compounds such as hydrogen sulfide and sulfur dioxide (SO2) may be derived from natural sources or from industrial air pollution.
| Dry air | |||
|---|---|---|---|
| Gas | Mole fraction(A) | ||
| Name | Formula | in ppm(B) | in % |
| Nitrogen | N2 | 780,840 | 78.084 |
| Oxygen | O2 | 209,460 | 20.946 |
| Argon | Ar | 9,340 | 0.9340 |
| Carbon dioxide (April 2022)(C)[13] |
CO2 | 417 | 0.0417 |
| Neon | Ne | 18.18 | 0.001818 |
| Helium | He | 5.24 | 0.000524 |
| Methane (2022)(C)[14] |
CH4 | 1.91 | 0.000191 |
| Krypton | Kr | 1.14 | 0.000114 |
| If air is not dry: | |||
| Water vapor(D) | H2O | 0–30,000(D) | 0–3%(E) |
notes:
| |||
The average molecular weight of dry air, which can be used to calculate densities or to convert between mole fraction and mass fraction, is about 28.946[15] or 28.96[16][17] g/mol. This is decreased when the air is humid.
The relative concentration of gases remains constant until about 10,000 m (33,000 ft).[18]
Stratification

In general, air pressure and density decrease with altitude in the atmosphere. However, the temperature has a more complicated profile with altitude, and may remain relatively constant or even increase with altitude in some regions (see the temperature section, below). Because the general pattern of the temperature/altitude profile, or lapse rate, is constant and measurable by means of instrumented balloon soundings, the temperature behavior provides a useful metric to distinguish atmospheric layers. In this way, Earth's atmosphere can be divided (called atmospheric stratification) into five main layers: troposphere, stratosphere, mesosphere, thermosphere, and exosphere.[19] The altitudes of the five layers are as follows:
- Exosphere: 700 to 10,000 km (440 to 6,200 miles)[20]
- Thermosphere: 80 to 700 km (50 to 440 miles)[21]
- Mesosphere: 50 to 80 km (31 to 50 miles)
- Stratosphere: 12 to 50 km (7 to 31 miles)
- Troposphere: 0 to 12 km (0 to 7 miles)[22]
Exosphere
The exosphere is the outermost layer of Earth's atmosphere (though it is so tenuous that some scientists consider it to be part of interplanetary space rather than part of the atmosphere). It extends from the thermopause (also known as the "exobase") at the top of the thermosphere to a poorly defined boundary with the solar wind and interplanetary medium. The altitude of the exobase varies from about 500 kilometres (310 mi; 1,600,000 ft) to about 1,000 kilometres (620 mi) in times of higher incoming solar radiation.[23]
The upper limit varies depending on the definition. Various authorities consider it to end at about 10,000 kilometres (6,200 mi)[24] or about 190,000 kilometres (120,000 mi)—about halfway to the moon, where the influence of Earth's gravity is about the same as radiation pressure from sunlight.[23] The geocorona visible in the far ultraviolet (caused by neutral hydrogen) extends to at least 100,000 kilometres (62,000 mi).[23]
This layer is mainly composed of extremely low densities of hydrogen, helium and several heavier molecules including nitrogen, oxygen and carbon dioxide closer to the exobase. The atoms and molecules are so far apart that they can travel hundreds of kilometres without colliding with one another. Thus, the exosphere no longer behaves like a gas, and the particles constantly escape into space. These free-moving particles follow ballistic trajectories and may migrate in and out of the magnetosphere or the solar wind. Every second, the Earth loses about 3 kg of hydrogen, 50 g of helium, and much smaller amounts of other constituents.[25]
The exosphere is too far above Earth for meteorological phenomena to be possible. However, Earth's auroras—the aurora borealis (northern lights) and aurora australis (southern lights)—sometimes occur in the lower part of the exosphere, where they overlap into the thermosphere. The exosphere contains many of the artificial satellites that orbit Earth.
Thermosphere
The thermosphere is the second-highest layer of Earth's atmosphere. It extends from the mesopause (which separates it from the mesosphere) at an altitude of about 80 km (50 mi; 260,000 ft) up to the thermopause at an altitude range of 500–1000 km (310–620 mi; 1,600,000–3,300,000 ft). The height of the thermopause varies considerably due to changes in solar activity.[21] Because the thermopause lies at the lower boundary of the exosphere, it is also referred to as the exobase. The lower part of the thermosphere, from 80 to 550 kilometres (50 to 342 mi) above Earth's surface, contains the ionosphere.
The temperature of the thermosphere gradually increases with height and can rise as high as 1500 °C (2700 °F), though the gas molecules are so far apart that its temperature in the usual sense is not very meaningful. The air is so rarefied that an individual molecule (of oxygen, for example) travels an average of 1 kilometre (0.62 mi; 3300 ft) between collisions with other molecules.[26] Although the thermosphere has a high proportion of molecules with high energy, it would not feel hot to a human in direct contact, because its density is too low to conduct a significant amount of energy to or from the skin.
This layer is completely cloudless and free of water vapor. However, non-hydrometeorological phenomena such as the aurora borealis and aurora australis are occasionally seen in the thermosphere. The International Space Station orbits in this layer, between 350 and 420 km (220 and 260 mi). It is this layer where many of the satellites orbiting the Earth are present.
Mesosphere

The mesosphere is the third highest layer of Earth's atmosphere, occupying the region above the stratosphere and below the thermosphere. It extends from the stratopause at an altitude of about 50 km (31 mi; 160,000 ft) to the mesopause at 80–85 km (50–53 mi; 260,000–280,000 ft) above sea level.
Temperatures drop with increasing altitude to the mesopause that marks the top of this middle layer of the atmosphere. It is the coldest place on Earth and has an average temperature around −85 °C (−120 °F; 190 K).[27][28]
Just below the mesopause, the air is so cold that even the very scarce water vapor at this altitude can condense into polar-mesospheric noctilucent clouds of ice particles. These are the highest clouds in the atmosphere and may be visible to the naked eye if sunlight reflects off them about an hour or two after sunset or similarly before sunrise. They are most readily visible when the Sun is around 4 to 16 degrees below the horizon. Lightning-induced discharges known as transient luminous events (TLEs) occasionally form in the mesosphere above tropospheric thunderclouds. The mesosphere is also the layer where most meteors burn up upon atmospheric entrance. It is too high above Earth to be accessible to jet-powered aircraft and balloons, and too low to permit orbital spacecraft. The mesosphere is mainly accessed by sounding rockets and rocket-powered aircraft.
Stratosphere
The stratosphere is the second-lowest layer of Earth's atmosphere. It lies above the troposphere and is separated from it by the tropopause. This layer extends from the top of the troposphere at roughly 12 km (7.5 mi; 39,000 ft) above Earth's surface to the stratopause at an altitude of about 50 to 55 km (31 to 34 mi; 164,000 to 180,000 ft).
The atmospheric pressure at the top of the stratosphere is roughly 1/1000 the pressure at sea level. It contains the ozone layer, which is the part of Earth's atmosphere that contains relatively high concentrations of that gas. The stratosphere defines a layer in which temperatures rise with increasing altitude. This rise in temperature is caused by the absorption of ultraviolet radiation (UV) from the Sun by the ozone layer, which restricts turbulence and mixing. Although the temperature may be −60 °C (−76 °F; 210 K) at the tropopause, the top of the stratosphere is much warmer, and may be near 0 °C.[29]
The stratospheric temperature profile creates very stable atmospheric conditions, so the stratosphere lacks the weather-producing air turbulence that is so prevalent in the troposphere. Consequently, the stratosphere is almost completely free of clouds and other forms of weather. However, polar stratospheric or nacreous clouds are occasionally seen in the lower part of this layer of the atmosphere where the air is coldest. The stratosphere is the highest layer that can be accessed by jet-powered aircraft.
Troposphere

The troposphere is the lowest layer of Earth's atmosphere. It extends from Earth's surface to an average height of about 12 km (7.5 mi; 39,000 ft), although this altitude varies from about 9 km (5.6 mi; 30,000 ft) at the geographic poles to 17 km (11 mi; 56,000 ft) at the Equator,[22] with some variation due to weather. The troposphere is bounded above by the tropopause, a boundary marked in most places by a temperature inversion (i.e. a layer of relatively warm air above a colder one), and in others by a zone that is isothermal with height.[30][31]
Although variations do occur, the temperature usually declines with increasing altitude in the troposphere because the troposphere is mostly heated through energy transfer from the surface. Thus, the lowest part of the troposphere (i.e. Earth's surface) is typically the warmest section of the troposphere. This promotes vertical mixing (hence, the origin of its name in the Greek word τρόπος, tropos, meaning "turn"). The troposphere contains roughly 80% of the mass of Earth's atmosphere.[32] The troposphere is denser than all its overlying layers because a larger atmospheric weight sits on top of the troposphere and causes it to be most severely compressed. Fifty percent of the total mass of the atmosphere is located in the lower 5.6 km (3.5 mi; 18,000 ft) of the troposphere.
Nearly all atmospheric water vapor or moisture is found in the troposphere, so it is the layer where most of Earth's weather takes place. It has basically all the weather-associated cloud genus types generated by active wind circulation, although very tall cumulonimbus thunder clouds can penetrate the tropopause from below and rise into the lower part of the stratosphere. Most conventional aviation activity takes place in the troposphere, and it is the only layer accessible by propeller-driven aircraft.
Other layers
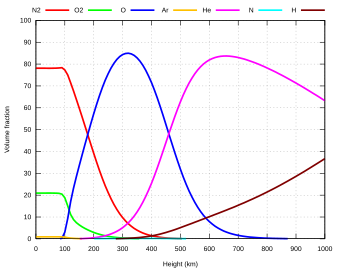
Within the five principal layers above, which are largely determined by temperature, several secondary layers may be distinguished by other properties:
- The ozone layer is contained within the stratosphere. In this layer ozone concentrations are about 2 to 8 parts per million, which is much higher than in the lower atmosphere but still very small compared to the main components of the atmosphere. It is mainly located in the lower portion of the stratosphere from about 15–35 km (9.3–21.7 mi; 49,000–115,000 ft), though the thickness varies seasonally and geographically. About 90% of the ozone in Earth's atmosphere is contained in the stratosphere.
- The ionosphere is a region of the atmosphere that is ionized by solar radiation. It is responsible for auroras. During daytime hours, it stretches from 50 to 1,000 km (31 to 621 mi; 160,000 to 3,280,000 ft) and includes the mesosphere, thermosphere, and parts of the exosphere. However, ionization in the mesosphere largely ceases during the night, so auroras are normally seen only in the thermosphere and lower exosphere. The ionosphere forms the inner edge of the magnetosphere. It has practical importance because it influences, for example, radio propagation on Earth.
- The homosphere and heterosphere are defined by whether the atmospheric gases are well mixed. The surface-based homosphere includes the troposphere, stratosphere, mesosphere, and the lowest part of the thermosphere, where the chemical composition of the atmosphere does not depend on molecular weight because the gases are mixed by turbulence.[33] This relatively homogeneous layer ends at the turbopause found at about 100 km (62 mi; 330,000 ft), the very edge of space itself as accepted by the FAI, which places it about 20 km (12 mi; 66,000 ft) above the mesopause.
- Above this altitude lies the heterosphere, which includes the exosphere and most of the thermosphere. Here, the chemical composition varies with altitude. This is because the distance that particles can move without colliding with one another is large compared with the size of motions that cause mixing. This allows the gases to stratify by molecular weight, with the heavier ones, such as oxygen and nitrogen, present only near the bottom of the heterosphere. The upper part of the heterosphere is composed almost completely of hydrogen, the lightest element.[34]
- The planetary boundary layer is the part of the troposphere that is closest to Earth's surface and is directly affected by it, mainly through turbulent diffusion. During the day the planetary boundary layer usually is well-mixed, whereas at night it becomes stably stratified with weak or intermittent mixing. The depth of the planetary boundary layer ranges from as little as about 100 metres (330 ft) on clear, calm nights to 3,000 m (9,800 ft) or more during the afternoon in dry regions.
The average temperature of the atmosphere at Earth's surface is 14 °C (57 °F; 287 K)[35] or 15 °C (59 °F; 288 K),[36] depending on the reference.[37][38][39]
Physical properties
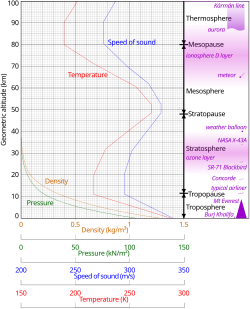
Pressure and thickness
The average atmospheric pressure at sea level is defined by the International Standard Atmosphere as 101325 pascals (760.00 Torr; 14.6959 psi; 760.00 mmHg). This is sometimes referred to as a unit of standard atmospheres (atm). Total atmospheric mass is 5.1480×1018 kg (1.135×1019 lb),[41] about 2.5% less than would be inferred from the average sea level pressure and Earth's area of 51007.2 megahectares, this portion being displaced by Earth's mountainous terrain. Atmospheric pressure is the total weight of the air above unit area at the point where the pressure is measured. Thus air pressure varies with location and weather.
If the entire mass of the atmosphere had a uniform density equal to sea level density (about 1.2 kg per m3) from sea level upwards, it would terminate abruptly at an altitude of 8.50 km (27,900 ft).
Air pressure actually decreases exponentially with altitude, dropping by half every 5.6 km (18,000 ft) or by a factor of 1/e (0.368) every 7.64 km (25,100 ft), (this is called the scale height) -- for altitudes out to around 70 km (43 mi; 230,000 ft). However, the atmosphere is more accurately modeled with a customized equation for each layer that takes gradients of temperature, molecular composition, solar radiation and gravity into account. At heights over 100 km, an atmosphere may no longer be well mixed. Then each chemical species has its own scale height.
In summary, the mass of Earth's atmosphere is distributed approximately as follows:[42]
- 50% is below 5.6 km (18,000 ft).
- 90% is below 16 km (52,000 ft).
- 99.99997% is below 100 km (62 mi; 330,000 ft), the Kármán line. By international convention, this marks the beginning of space where human travelers are considered astronauts.
By comparison, the summit of Mount Everest is at 8,848 m (29,029 ft); commercial airliners typically cruise between 10 and 13 km (33,000 and 43,000 ft) where the lower density and temperature of the air improve fuel economy; weather balloons reach 30.4 km (100,000 ft) and above; and the highest X-15 flight in 1963 reached 108.0 km (354,300 ft).
Even above the Kármán line, significant atmospheric effects such as auroras still occur. Meteors begin to glow in this region, though the larger ones may not burn up until they penetrate more deeply. The various layers of Earth's ionosphere, important to HF radio propagation, begin below 100 km and extend beyond 500 km. By comparison, the International Space Station and Space Shuttle typically orbit at 350–400 km, within the F-layer of the ionosphere where they encounter enough atmospheric drag to require reboosts every few months, otherwise, orbital decay will occur resulting in a return to Earth. Depending on solar activity, satellites can experience noticeable atmospheric drag at altitudes as high as 700–800 km.
Temperature
The division of the atmosphere into layers mostly by reference to temperature is discussed above. Temperature decreases with altitude starting at sea level, but variations in this trend begin above 11 km, where the temperature stabilizes over a large vertical distance through the rest of the troposphere. In the stratosphere, starting above about 20 km, the temperature increases with height, due to heating within the ozone layer caused by the capture of significant ultraviolet radiation from the Sun by the dioxygen and ozone gas in this region. Still another region of increasing temperature with altitude occurs at very high altitudes, in the aptly-named thermosphere above 90 km.
Speed of sound
Because in an ideal gas of constant composition the speed of sound depends only on temperature and not on pressure or density, the speed of sound in the atmosphere with altitude takes on the form of the complicated temperature profile (see illustration to the right), and does not mirror altitudinal changes in density or pressure.
Density and mass

The density of air at sea level is about 1.2 kg/m3 (1.2 g/L, 0.0012 g/cm3). Density is not measured directly but is calculated from measurements of temperature, pressure and humidity using the equation of state for air (a form of the ideal gas law). Atmospheric density decreases as the altitude increases. This variation can be approximately modeled using the barometric formula. More sophisticated models are used to predict the orbital decay of satellites.
The average mass of the atmosphere is about 5 quadrillion (5×1015) tonnes or 1/1,200,000 the mass of Earth. According to the American National Center for Atmospheric Research, "The total mean mass of the atmosphere is 5.1480×1018 kg with an annual range due to water vapor of 1.2 or 1.5×1015 kg, depending on whether surface pressure or water vapor data are used; somewhat smaller than the previous estimate. The mean mass of water vapor is estimated as 1.27×1016 kg and the dry air mass as 5.1352 ±0.0003×1018 kg."
Tabulated properties
Table of physical and thermal properties of air at atmospheric pressure:[44][45]
| Temperature | Density | Specific heat | Dynamic viscosity | Kinematic viscosity | Thermal conductivity | Thermal diffusivity | Prandtl Number | Bulk modulus |
| 100 | 3.601 | 1026.6 | 6.92×10−06 | 1.92×10−06 | 0.000925 | 2.50×10−06 | 0.77 | 0.01 |
| 150 | 2.3675 | 1009.9 | 1.03×10−05 | 4.34×10−06 | 0.013735 | 5.75×10−6 | 0.753 | 0.006667 |
| 200 | 1.7684 | 1006.1 | 1.33×10−05 | 7.49×10−06 | 0.01809 | 1.02×10−05 | 0.738 | 0.005 |
| 250 | 1.4128 | 1005.3 | 1.60×10−05 | 1.13×10−05 | 0.02227 | 1.57×10−05 | 0.722 | 0.004 |
| 300 | 1.1774 | 1005.7 | 1.85×10−05 | 1.57×10−05 | 0.02624 | 2.22×10−05 | 0.708 | 0.003333 |
| 350 | 0.998 | 1009 | 2.08×10−05 | 2.08×10−05 | 0.03003 | 2.98×10−05 | 0.697 | 0.002857 |
| 400 | 0.8826 | 1014 | 2.29×10−05 | 2.59×10−05 | 0.03365 | 3.76×10−05 | 0.689 | 0.0025 |
| 450 | 0.7833 | 1020.7 | 2.48×10−05 | 3.17×10−05 | 0.03707 | 4.22×10−05 | 0.683 | 0.002222 |
| 500 | 0.7048 | 1029.5 | 2.67×10−05 | 3.79×10−05 | 0.04038 | 5.56×10−05 | 0.68 | 0.002 |
| 550 | 0.6423 | 1039.2 | 2.85×10−05 | 4.43×10−05 | 0.0436 | 6.53×10−05 | 0.68 | 0.001818 |
| 600 | 0.5879 | 1055.1 | 3.02×10−05 | 5.13×10−05 | 0.04659 | 7.51×10−05 | 0.68 | 0.001667 |
| 650 | 0.543 | 1063.5 | 3.18×10−05 | 5.85×10−05 | 0.04953 | 8.58×10−05 | 0.682 | 0.001538 |
| 700 | 0.503 | 1075.2 | 3.33×10−05 | 6.63×10−05 | 0.0523 | 9.67×10−05 | 0.684 | 0.001429 |
| 750 | 0.4709 | 1085.6 | 3.48×10−05 | 7.39×10−05 | 0.05509 | 1.08×10−04 | 0.686 | 0.001333 |
| 800 | 0.4405 | 1097.8 | 3.63×10−05 | 8.23×10−05 | 0.05779 | 1.20×10−04 | 0.689 | 0.00125 |
| 850 | 0.4149 | 1109.5 | 3.77×10−05 | 9.08×10−05 | 0.06028 | 1.31×10−04 | 0.692 | 0.001176 |
| 900 | 0.3925 | 1121.2 | 3.90×10−05 | 9.93×10−05 | 0.06279 | 1.43×10−04 | 0.696 | 0.001111 |
| 950 | 0.3716 | 1132.1 | 4.02×10−05 | 1.08×10−04 | 0.06525 | 1.55×10−04
Zdroj:https://en.wikipedia.org?pojem=Atmosphere_of_Earth Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Analytika
Antropológia Aplikované vedy Bibliometria Dejiny vedy Encyklopédie Filozofia vedy Forenzné vedy Humanitné vedy Knižničná veda Kryogenika Kryptológia Kulturológia Literárna veda Medzidisciplinárne oblasti Metódy kvantitatívnej analýzy Metavedy Metodika Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok. www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk |
