A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Carbon dioxide
| |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3DMet | |||
| 1900390 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.271 | ||
| EC Number |
| ||
| E number | E290 (preservatives) | ||
| 989 | |||
| KEGG | |||
| MeSH | Carbon+dioxide | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1013 (gas), 1845 (solid) | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CO2 | |||
| Molar mass | 44.009 g·mol−1 | ||
| Appearance | Colorless gas | ||
| Odor |
| ||
| Density |
| ||
| Critical point (T, P) | 304.128(15) K[2] (30.978(15) °C), 7.3773(30) MPa[2] (72.808(30) atm) | ||
| 194.6855(30) K (−78.4645(30) °C) at 1 atm (0.101325 MPa) | |||
| 1.45 g/L at 25 °C (77 °F), 100 kPa (0.99 atm) | |||
| Vapor pressure | 5.7292(30) MPa, 56.54(30) atm (20 °C (293.15 K)) | ||
| Acidity (pKa) | Carbonic acid: pKa1 = 3.6 pKa1(apparent) = 6.35 pKa2 = 10.33 | ||
| −20.5·10−6 cm3/mol | |||
| Thermal conductivity | 0.01662 W·m−1·K−1 (300 K (27 °C; 80 °F))[3] | ||
Refractive index (nD)
|
1.00045 | ||
| Viscosity |
| ||
| 0 D | |||
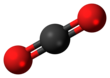
| Structure | |||
| Trigonal | |||
| Linear | |||
| Thermochemistry | |||
Heat capacity (C)
|
37.135 J/(K·mol) | ||
Std molar
entropy (S⦵298) |
214 J·mol−1·K−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
−393.5 kJ·mol−1 | ||
| Pharmacology | |||
| V03AN02 (WHO) | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LCLo (lowest published)
|
90,000 ppm (162,000 mg/m3) (human, 5 min)[6] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 5000 ppm (9000 mg/m3)[5] | ||
REL (Recommended)
|
TWA 5000 ppm (9000 mg/m3), ST 30,000 ppm (54,000 mg/m3)[5] | ||
IDLH (Immediate danger)
|
40,000 ppm (72,000 mg/m3)[5] | ||
| Safety data sheet (SDS) | Sigma-Aldrich | ||
| Related compounds | |||
Other anions
|
|||