
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
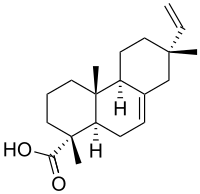
| IUPAC name
(13S)-Pimara-7,15-dien-18-oic acid
| |
| Systematic IUPAC name
(1R,4aR,4bS,7S,10aR)-7-Ethenyl-1,4a,7-trimethyl-1,2,3,4,4a,4b,5,6,7,8,10,10a-dodecahydrophenanthrene-1-carboxylic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.163.144 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H30O2 | |
| Molar mass | 302.458 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Isopimaric acid (IPA) is a toxin which acts as a large conductance Ca2+-activated K+ channel (BK channel) opener.
Sources
IPA originates from many sorts of trees, especially conifers.[1]
Chemistry
IPA is one of the members of the resin acid group and it is a tricyclic diterpene.[1]
Target
IPA acts on the large-conductance calcium activated K+ channels (BK channels).[2][3]
Mode of action
BK channels are formed by α subunits and accessory β subunits arranged in tetramers. The α subunit forms the ion conduction pore and the β subunit contributes to channel gating. IPA interaction with the BK channel enhances Ca2+ and / or voltage sensitivity of the α subunit of BK channels without affecting the channel conductance. In this state BK channels can still be inhibited by one of their inhibitors, like charybdotoxin (CTX).[2][3] Opening of the BK channel leads to an increased K+-efflux which hyperpolarizes the resting membrane potential, reducing the excitability of the cell in which the BK-channel is expressed.
Toxicity
Studies on rainbow trout hepatocytes have shown that IPA increases intracellular calcium release, leading to a disturbance in the calcium homeostasis. This could be important in the possible toxicity of the toxin.
See also
Notes
- ^ a b Wilson, AE; Moore, ER; Mohn, WW (1996). "Isolation and characterization of isopimaric acid-degrading bacteria from a sequencing batch reactor". Applied and Environmental Microbiology. 62 (9): 3146–51. Bibcode:1996ApEnM..62.3146W. doi:10.1128/aem.62.9.3146-3151.1996. PMC 168108. PMID 8795202.
- ^ a b Kaczorowski, GJ; Knaus, HG; Leonard, RJ; McManus, OB; Garcia, ML (1996). "High-conductance calcium-activated potassium channels; structure, pharmacology, and function". Journal of Bioenergetics and Biomembranes. 28 (3): 255–67. doi:10.1007/bf02110699. PMID 8807400. S2CID 25857254.
- ^ a b Imaizumi, Y; Sakamoto, K; Yamada, A; Hotta, A; Ohya, S; Muraki, K; Uchiyama, M; Ohwada, T (2002). "Molecular basis of pimarane compounds as novel activators of large-conductance Ca(2+)-activated K(+) channel alpha-subunit". Molecular Pharmacology. 62 (4): 836–46. doi:10.1124/mol.62.4.836. PMID 12237330.
References
- Råbergh, Christina M.I.; Lilius, Henrik; Eriksson, John E.; Isomaa, Boris (1999). "The resin acids dehydroabietic acid and isopimaric acid release calcium from intracellular stores in rainbow trout hepatocytes". Aquatic Toxicology. 46: 55–65. doi:10.1016/S0166-445X(98)00115-5.
- Råbergh, C.M.I.; Isomaa, B.; Eriksson, J.E. (1992). "The resin acids dehydroabietic acid and isopimaric acid inhibit bile acid uptake and perturb potassium transport in isolated hepatocvtes from rainbow trout (Oncorhynchus mykiss)". Aquatic Toxicology. 23 (3–4): 169–179. doi:10.1016/0166-445X(92)90050-W.
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
