
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| Atomic clock | |
|---|---|
 NIST physicists Steve Jefferts (foreground) and Tom Heavner with the NIST-F2 caesium fountain atomic clock, a civilian time standard for the United States | |
| Classification | Clock |
| Industry | Telecommunications, science |
| Application | TAI, satellite navigation |
| Fuel source | Electricity |
| Powered | Yes |

An atomic clock is a clock that measures time by monitoring the resonant frequency of atoms. It is based on atoms having different energy levels. Electron states in an atom are associated with different energy levels, and in transitions between such states they interact with a very specific frequency of electromagnetic radiation. This phenomenon serves as the basis for the International System of Units' (SI) definition of a second:
The second, symbol s, is the SI unit of time. It is defined by taking the fixed numerical value of the caesium frequency, , the unperturbed ground-state hyperfine transition frequency of the caesium-133 atom, to be 9192631770 when expressed in the unit Hz, which is equal to s−1.
This definition is the basis for the system of International Atomic Time (TAI), which is maintained by an ensemble of atomic clocks around the world. The system of Coordinated Universal Time (UTC) that is the basis of civil time implements leap seconds to allow clock time to track changes in Earth's rotation to within one second while being based on clocks that are based on the definition of the second.
The accurate timekeeping capabilities of atomic clocks are also used for navigation by satellite networks such as the European Union's Galileo Programme and the United States' GPS. The timekeeping accuracy of the involved atomic clocks is important because the smaller the error in time measurement, the smaller the error in distance obtained by multiplying the time by the speed of light is (a timing error of a nanosecond or 1 billionth of a second (10−9 or 1⁄1,000,000,000 second) translates into an almost 30-centimetre (11.8 in) distance and hence positional error).
The main variety of atomic clock uses caesium atoms cooled to temperatures that approach absolute zero. The primary standard for the United States, the National Institute of Standards and Technology (NIST)'s caesium fountain clock named NIST-F2, measures time with an uncertainty of 1 second in 300 million years (relative uncertainty 10−16). NIST-F2 was brought online on 3 April 2014.[2][3]
History

The Scottish physicist James Clerk Maxwell proposed measuring time with the vibrations of light waves in his 1873 Treatise on Electricity and Magnetism: 'A more universal unit of time might be found by taking the periodic time of vibration of the particular kind of light whose wave length is the unit of length.'[4][5] Maxwell argued this would be more accurate than the Earth's rotation, which defines the mean solar second for timekeeping.[6]
During the 1930s, the American physicist Isidor Isaac Rabi built equipment for atomic beam magnetic resonance frequency clocks.[7][8]
The accuracy of mechanical, electromechanical and quartz clocks is reduced by temperature fluctuations. This led to the idea of measuring the frequency of an atom's vibrations to keep time much more accurately, as proposed by James Clerk Maxwell, Lord Kelvin, and Isidor Rabi.[9] He proposed the concept in 1945, which led to a demonstration of a clock based on ammonia in 1949.[10] This led to the first practical accurate atomic clock with caesium atoms being built at the National Physical Laboratory in the United Kingdom in 1955[11][12] by Louis Essen in collaboration with Jack Parry.[13]

In 1949, Kastler and Brossel [15] developed a technique called optical pumping for electron energy level transitions in atoms using light. This technique is useful for creating much stronger magnetic resonance and microwave absorption signals. Unfortunately, this caused a side effect with a light shift of the resonant frequency. Cohen-Tannoudji and others managed to reduce the light shifts to acceptable levels.
Ramsey developed a method, commonly known as Ramsey interferometry nowadays, for higher frequencies and narrower resonances in the oscillating fields. Kolsky, Phipps, Ramsey, and Silsbee used this technique for molecular beam spectroscopy in 1950.[16]
After 1956, atomic clocks were studied by many groups, such as the National Institute of Standards and Technology (formerly the National Bureau of Standards) in the USA, the Physikalisch-Technische Bundesanstalt (PTB) in Germany, the National Research Council (NRC) in Canada, the National Physical Laboratory in the United Kingdom, International Time Bureau (French: Bureau International de l'Heure, abbreviated BIH), at the Paris Observatory, the National Radio Company, Bomac, Varian, Hewlett–Packard and Frequency & Time Systems.[17]
During the 1950s, the National Radio Company sold more than 50 units of the first atomic clock, the Atomichron.[18] In 1964, engineers at Hewlett-Packard released the 5060 rack-mounted model of caesium clocks.[9]
Definition of the second
In 1968, the duration of the second was defined to be 9192631770 vibrations of the unperturbed ground-state hyperfine transition frequency of the caesium-133 atom. Prior to that it was defined by there being 31556925.9747 seconds in the tropical year 1900.[19] The 1968 definition was updated in 2019 to reflect the new definitions of the ampere, kelvin, kilogram, and mole decided upon at the 2019 redefinition of the International System of Units. Timekeeping researchers are currently working on developing an even more stable atomic reference for the second, with a plan to find a more precise definition of the second as atomic clocks improve based on optical clocks or the Rydberg constant around 2030.[20][21]
Metrology advancements and optical clocks

Technological developments such as lasers and optical frequency combs in the 1990s led to increasing accuracy of atomic clocks.[22][23] Lasers enable the possibility of optical-range control over atomic states transitions, which has a much higher frequency than that of microwaves; while optical frequency comb measures highly accurately such high frequency oscillation in light.
The first advance beyond the precision of caesium clocks occurred at NIST in 2010 with the demonstration of a "quantum logic" optical clock that used aluminum ions to achieve a precision of 10−17.[24] Optical clocks are a very active area of research in the field of metrology as scientists work to develop clocks based on elements ytterbium, mercury, aluminum, and strontium. Scientists at JILA demonstrated a strontium clock with a frequency precision of 10−18 in 2015.[25] Scientists at NIST developed a quantum logic clock that measured a single aluminum ion in 2019 with a frequency uncertainty of 10−19.[26][27]
At JILA in September 2021, scientists demonstrated an optical strontium clock with a frequency differential precision of 7.6×10−21.[28][29] The second is expected to be redefined when the field of optical clocks matures, sometime around the year 2026 or 2030.[21] In order for this to occur, optical clocks must be capable of measuring time to very high precision consistently. In addition, methods for reliably and accurately comparing different optical clocks around the world in national metrology labs must be demonstrated.
Chip-scale atomic clocks

In addition to increased accuracy, the development of chip-scale atomic clocks has expanded the number of places atomic clocks can be used. In August 2004, NIST scientists demonstrated a chip-scale atomic clock that was 100 times smaller than an ordinary atomic clock and had a much smaller power consumption of 125 mW.[30][31] The atomic clock was about the size of a grain of rice with a frequency of about 9 GHz. This technology became available commercially in 2011.[30] Atomic clocks on the scale of one chip require less than 30 milliwatts of power.[32][33]
The National Institute of Standards and Technology created a program NIST on a chip to develop compact ways of measuring time with a device just a few millimeters across.[34]
Metrologists are currently (2022) designing atomic clocks that implement new developments such as ion traps and optical combs to reach greater accuracies.[35]
How atomic clocks work
Time standards
An atomic clock is based on a system of atoms which may be in one of two possible energy states. A group of atoms in one state is prepared, then subjected to microwave radiation. If the radiation is of the correct frequency, a number of atoms will transition to the other energy state. The closer the frequency is to the inherent oscillation frequency of the atoms, the more atoms will switch states. Such correlation allows very accurate tuning of the frequency of the microwave radiation. Once the microwave radiation is adjusted to a known frequency where the maximum number of atoms switch states, the atom and thus, its associated transition frequency, can be used as a timekeeping oscillator to measure elapsed time.[36]
A number of national metrology laboratories maintain atomic clocks: including Paris Observatory, the Physikalisch-Technische Bundesanstalt (PTB) in Germany, the National Institute of Standards and Technology (NIST) in Colorado and Maryland, USA, JILA in the University of Colorado Boulder, the National Physical Laboratory (NPL) in the United Kingdom, and the All-Russian Scientific Research Institute for Physical-Engineering and Radiotechnical Metrology. They do this by designing and building frequency standards that produce electric oscillations at a frequency whose relationship to the transition frequency of caesium 133 is known, in order to achieve a very low uncertainty. These primary frequency standards estimate and correct various frequency shifts, including relativistic Doppler shifts linked to atomic motion, the thermal radiation of the environment (blackbody shift) and several other factors. The best primary standards currently produce the SI second with an accuracy approaching an uncertainty of one part in 1016.
It is important to note that at this level of accuracy, the differences in the gravitational field in the device cannot be ignored. The standard is then considered in the framework of general relativity to provide a proper time at a specific point.[37]
The International Bureau of Weights and Measures (BIPM) provides a list of frequencies that serve as secondary representations of the second. This list contains the frequency values and respective standard uncertainties for the rubidium microwave transition and other optical transitions, including neutral atoms and single trapped ions. These secondary frequency standards can be as accurate as one part in 1018; however, the uncertainties in the list are one part in 1014–1016. This is because the uncertainty in the central caesium standard against which the secondary standards are calibrated is one part in 1014–1016.
Primary frequency standards can be used to calibrate the frequency of other clocks used in national laboratories. These are usually commercial caesium clocks having very good long-term frequency stability, maintaining a frequency with a stability better than 1 part in 1014 over a few months. The uncertainty of the primary standard frequencies is around one part in 1013.
Hydrogen masers, which rely on the 1.4 GHz hyperfine transition in atomic hydrogen, are also used in time metrology laboratories. Masers outperform any commercial caesium clock in terms of short-term frequency stability. In the past, these instruments have been used in all applications that require a steady reference across time periods of less than one day (frequency stability of about 1 part in ten[clarification needed] for averaging times of a few hours). Because some active hydrogen masers have a modest but predictable frequency drift with time, they have become an important part of the BIPM's ensemble of commercial clocks that implement International Atomic Time.[37]
Synchronization with satellites
The time readings of clocks operated in metrology labs operating with the BIPM need to be known very accurately. Some operations require synchronization of atomic clocks separated by great distances over thousands of kilometers. Global Navigational Satellite Systems (GNSS) provide a satisfactory solution to the problem of time transfer. Atomic clocks are used to broadcast time signals in the United States Global Positioning System (GPS), the Russian Federation's Global Navigation Satellite System (GLONASS), the European Union's Galileo system and China's BeiDou system.
The signal received from one satellite in a metrology laboratory equipped with a receiver with an accurately known position allows the time difference between the local time scale and the GNSS system time to be determined with an uncertainty of a few nanoseconds when averaged over 15 minutes. Receivers allow the simultaneous reception of signals from several satellites, and make use of signals transmitted on two frequencies. As more satellites are launched and start operations, time measurements will become more accurate.
These methods of time comparison must make corrections for the effects of special relativity and general relativity of a few nanoseconds.
International timekeeping

National laboratories usually operate a range of clocks. These are operated independently of one another and their measurements are sometimes combined to generate a scale that is more stable and more accurate than that of any individual contributing clocks. This scale allows for time comparisons between different clocks in the laboratory. These atomic time scales are generally referred to as TA(k) for laboratory k.[38]
Coordinated Universal Time (UTC) is the result of comparing clocks in national laboratories around the world to International Atomic Time (TAI), then adding leap seconds as necessary. TAI is a weighted average of around 450 clocks in some 80 time institutions.[39] The relative stability of TAI is around one part in 1016.
Before TAI is published, the frequency of the result is compared with the SI second at various primary and secondary frequency standards. This requires relativistic corrections to be applied to the location of the primary standard which depend on the distance between the equal gravity potential and the rotating geoid of Earth. The values of the rotating geoid and the TAI change slightly each month and are available in the BIPM Circular T publication. The TAI time-scale is deferred by a few weeks as the average of atomic clocks around the world is calculated.
TAI is not distributed in everyday timekeeping. Instead, an integer number of leap seconds are added or subtracted to correct for the Earth's rotation, producing UTC. The number of leap seconds is changed so that mean solar noon at the Greenwich meridian does not deviate from UTC noon by more than 0.9 seconds.
National metrology institutions maintain an approximation of UTC referred to as UTC(k) for laboratory k. UTC(k) is distributed by the BIPM's Consultative Committee for Time and Frequency. The offset UTC-UTC(k) is calculated every 5 days, the results are published monthly. Atomic clocks record UTC(k) to no more than 100 nanoseconds. In some countries, UTC(k) is the legal time that is distributed by radio, television, telephone, Internet, fiber-optic cables, time signal transmitters, and speaking clocks. In addition, GNSS provides time information accurate to a few tens of nanoseconds or better.
Types
Caesium
The SI second is defined as a certain number of unperturbed ground-state hyperfine transitions of the caesium-133 atom. Caesium standards are therefore regarded as primary time and frequency standards.
Caesium clocks include the NIST-F1 clock, developed in 1999, and the NIST-F2 clock, developed in 2013.[40][41]
Caesium has several properties that make it a good choice for an atomic clock. Whereas a hydrogen atom moves at 1,600 m/s at room temperature and a nitrogen atom moves at 510 m/s, a caesium atom moves at a much slower speed of 130 m/s due to its greater mass.[42][9] The hyperfine frequency of caesium (~9.19 GHz) is also higher than other elements such as rubidium (~6.8 GHz) and hydrogen (~1.4 GHz).[9] The high frequency of caesium allows for more accurate measurements. Caesium reference tubes suitable for national standards currently last about seven years and cost about US$35,000. Primary frequency and time standards like the United States Time Standard atomic clocks, NIST-F1 and NIST-F2, use far higher power.[31][43][44][45]
Block diagram
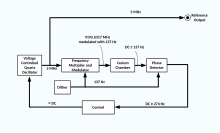
In a caesium beam frequency reference, timing signals are derived from a high stability voltage-controlled quartz crystal oscillator (VCXO) that is tunable over a narrow range. The output frequency of the VCXO (typically 5 MHz) is multiplied by a frequency synthesizer to obtain microwaves at the frequency of the caesium atomic hyperfine transition (about 9192.6317 MHz). The output of the frequency synthesizer is amplified and applied to a chamber containing caesium gas which absorbs the microwaves. The output current of the caesium chamber increases as absorption increases.
The remainder of the circuitry simply adjusts the running frequency of the VCXO to maximize the output current of the caesium chamber which keeps the oscillator tuned to the resonance frequency of the hyperfine transition.[46]
Rubidium

The BIPM defines the unperturbed ground-state hyperfine transition frequency of the rubidium-87 atom, 6 834 682 610.904 312 6 Hz, in terms of the caesium standard frequency. Atomic clocks based on rubidium standards are therefore regarded as secondary representations of the second.
Rubidium standard clocks are prized for their low cost, small size (commercial standards are as small as 1.7×105 mm3)[30] and short-term stability. They are used in many commercial, portable and aerospace applications. Modern rubidium standard tubes last more than ten years, and can cost as little as US$50. Some commercial applications use a rubidium standard periodically corrected by a global positioning system receiver (see GPS disciplined oscillator). This achieves excellent short-term accuracy, with long-term accuracy equal to (and traceable to) the US national time standards.[47]
Hydrogen

The BIPM defines the unperturbed optical transition frequency of the hydrogen-1 neutral atom, 1 233 030 706 593 514 Hz, in terms of the caesium standard frequency. Atomic clocks based on hydrogen standards are therefore regarded as secondary representations of the second.
Hydrogen masers have superior short-term stability compared to other standards, but lower long-term accuracy. The long-term stability of hydrogen maser standards decreases because of changes in the cavity's properties over time. The relative error of hydrogen masers is 5 × 10−16 for periods of 1000 seconds. This makes hydrogen masers good for radio astronomy, in particular for very long baseline interferometry.[5]
Hydrogen masers are used for flywheel oscillators in laser-cooled atomic frequency standards and broadcasting time signals from national standards laboratories, although they need to be corrected as they drift from the correct frequency over time. The hydrogen maser is also useful for experimental tests of the effects of special relativity and general relativity such as gravitational red shift.[5]
Strontium
This section needs expansion. You can help by adding to it. (August 2023) |
A few types of strontium atomic clocks exist, including optical clocks.
Time measuring mechanism
International System of Units definition
Since 1968, the SI has defined the second as the duration of 9192631770 cycles of radiation corresponding to the transition between two energy levels of the ground state of the caesium-133 atom. In 1997, the International Committee for Weights and Measures (CIPM) added that the preceding definition refers to a caesium atom at rest at a temperature of absolute zero.[48]: 113
This definition makes the caesium oscillator the primary standard for time and frequency measurements, called the caesium standard. Following the 2019 redefinition of the SI base units, the definition of every base unit except the mole and almost every derived unit relies on the definition of the second.
Tuning and optimization
The core of the traditional radio frequency atomic clock is a tunable microwave cavity containing a gas. In a hydrogen maser clock the gas emits microwaves (the gas mases) on a hyperfine transition, the field in the cavity oscillates, and the cavity is tuned for maximum microwave amplitude. Alternatively, in a caesium or rubidium clock, the beam or gas absorbs microwaves and the cavity contains an electronic amplifier to make it oscillate. For both types, the atoms in the gas are prepared in one hyperfine state prior to filling them into the cavity. For the second type, the number of atoms that change hyperfine state is detected and the cavity is tuned for a maximum of detected state changes.
Most of the complexity of the clock lies in this adjustment process. The adjustment tries to correct for unwanted side-effects, such as frequencies from other electron transitions, temperature changes, and the spreading in frequencies caused by the vibration of molecules including Doppler broadening.[49] One way of doing this is to sweep the microwave oscillator's frequency across a narrow range to generate a modulated signal at the detector. The detector's signal can then be demodulated to apply feedback to control long-term drift in the radio frequency.[50]
In this way, the quantum-mechanical properties of the atomic transition frequency of the caesium can be used to tune the microwave oscillator to the same frequency, except for a small amount of experimental error. When a clock is first turned on, it takes a while for the oscillator to stabilize. In practice, the feedback and monitoring mechanism is much more complex.[51]
Many of the newer clocks, including microwave clocks such as trapped ion or fountain clocks, and optical clocks such as lattice clocks use a sequential interrogation protocol rather than the frequency modulation interrogation described above.[52] An advantage of sequential interrogation is that it can accommodate much higher Q's, with ringing times of seconds rather than milliseconds. These clocks also typically have a dead time, during which the atom or ion collections are analyzed, renewed and driven into a proper quantum state, after which they are interrogated with a signal from a local oscillator (LO) for a time of perhaps a second or so. Analysis of the final state of the atoms is then used to generate a correction signal to keep the LO frequency locked to that of the atoms or ions.
Clock mechanism
All timekeeping devices use oscillatory phenomena to accurately measure time, whether it is the rotation of the Earth for a sundial, the swinging of a pendulum in a grandfather clock, the vibrations of springs and gears in a watch, or voltage changes in a quartz crystal watch. However all of these are easily affected by temperature changes and are not very accurate. The most accurate clocks use atomic vibrations to keep track of time. Clock transition states in atoms are insensitive to temperature and other environmental factors and the oscillation frequency is much higher than any of the other clocks (in microwave frequency regime and higher).
One of the most important factors in a clock's performance is the atomic line quality factor, Q, which is defined as the ratio of the absolute frequency of the resonance to the linewidth of the resonance itself . Atomic resonance has a much higher Q than mechanical devices. Atomic clocks can also be isolated from environmental effects to a much higher degree. Atomic clocks have the benefit that atoms are universal, which means that the oscillation frequency is also universal. This is different from quartz and mechanical time measurement devices that do not have a universal frequency.
A clock's quality can be specified by two parameters: accuracy and stability. Accuracy is a measurement of the degree to which the clock's ticking rate can be counted on to match some absolute standard such as the inherent hyperfine frequency of an isolated atom or ion. Stability describes how the clock performs when averaged over time to reduce the impact of noise and other short-term fluctuations (see precision).[53]
The instability of an atomic clock is specified by its Allan deviation .[54] The limiting instability due to atom or ion counting statistics is given by
where is the spectroscopic linewidth of the clock system, is the number of atoms or ions used in a single measurement, is the time required for one cycle, and is the averaging period. This means instability is smaller when the linewidth is smaller and when (the signal to noise ratio) is larger. The stability improves as the time over which the measurements are averaged increases from seconds to hours to days. The stability is most heavily affected by the oscillator frequency . This is why optical clocks such as strontium clocks (429 terahertz) are much more stable than caesium clocks (9.19 GHz).
Modern clocks such as atomic fountains or optical lattices that use sequential interrogation are found to generate type of noise that mimics and adds to the instability inherent in atom or ion counting. This effect is called the Dick effect[55] and is typically the primary stability limitation for the newer atomic clocks. It is an aliasing effect; high frequency noise components in the local oscillator ("LO") are heterodyned to near zero frequency by harmonics of the repeating variation in feedback sensitivity to the LO frequency. The effect places new and stringent requirements on the LO, which must now have low phase noise in addition to high stability, thereby increasing the cost and complexity of the system. For the case of an LO with Flicker frequency noise[56] where is independent of , the interrogation time is , and where the duty factor has typical values , the Allan deviation can be approximated as[57]
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk













