
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| Types of fats in food |
|---|
| Components |
| Manufactured fats |
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, from 4 to 28.[1] Fatty acids are a major component of the lipids (up to 70% by weight) in some species such as microalgae[2] but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important dietary sources of fuel for animals and important structural components for cells.
History
The concept of fatty acid (acide gras) was introduced in 1813 by Michel Eugène Chevreul,[3][4][5] though he initially used some variant terms: graisse acide and acide huileux ("acid fat" and "oily acid").[6]
Types of fatty acids

Fatty acids are classified in many ways: by length, by saturation vs unsaturation, by even vs odd carbon content, and by linear vs branched.
Length of fatty acids
- Short-chain fatty acids (SCFAs) are fatty acids with aliphatic tails of five or fewer carbons (e.g. butyric acid).[7]
- Medium-chain fatty acids (MCFAs) are fatty acids with aliphatic tails of 6 to 12[8] carbons, which can form medium-chain triglycerides.
- Long-chain fatty acids (LCFAs) are fatty acids with aliphatic tails of 13 to 21 carbons.[9]
- Very long chain fatty acids (VLCFAs) are fatty acids with aliphatic tails of 22 or more carbons.
Saturated fatty acids
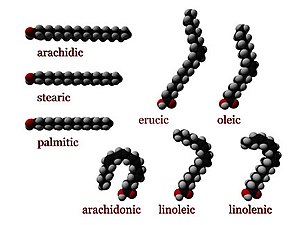
Saturated fatty acids have no C=C double bonds. They have the formula CH3(CH2)nCOOH, for different n. An important saturated fatty acid is stearic acid (n = 16), which when neutralized with sodium hydroxide is the most common form of soap.

| Common name | Chemical structure | C:D[a] |
|---|---|---|
| Caprylic acid | CH3(CH2)6COOH | 8:0 |
| Capric acid | CH3(CH2)8COOH | 10:0 |
| Lauric acid | CH3(CH2)10COOH | 12:0 |
| Myristic acid | CH3(CH2)12COOH | 14:0 |
| Palmitic acid | CH3(CH2)14COOH | 16:0 |
| Stearic acid | CH3(CH2)16COOH | 18:0 |
| Arachidic acid | CH3(CH2)18COOH | 20:0 |
| Behenic acid | CH3(CH2)20COOH | 22:0 |
| Lignoceric acid | CH3(CH2)22COOH | 24:0 |
| Cerotic acid | CH3(CH2)24COOH | 26:0 |
Unsaturated fatty acids
Unsaturated fatty acids have one or more C=C double bonds. The C=C double bonds can give either cis or trans isomers.
- cis
- A cis configuration means that the two hydrogen atoms adjacent to the double bond stick out on the same side of the chain. The rigidity of the double bond freezes its conformation and, in the case of the cis isomer, causes the chain to bend and restricts the conformational freedom of the fatty acid. The more double bonds the chain has in the cis configuration, the less flexibility it has. When a chain has many cis bonds, it becomes quite curved in its most accessible conformations. For example, oleic acid, with one double bond, has a "kink" in it, whereas linoleic acid, with two double bonds, has a more pronounced bend. α-Linolenic acid, with three double bonds, favors a hooked shape. The effect of this is that, in restricted environments, such as when fatty acids are part of a phospholipid in a lipid bilayer or triglycerides in lipid droplets, cis bonds limit the ability of fatty acids to be closely packed, and therefore can affect the melting temperature of the membrane or of the fat. Cis unsaturated fatty acids, however, increase cellular membrane fluidity, whereas trans unsaturated fatty acids do not.
- trans
- A trans configuration, by contrast, means that the adjacent two hydrogen atoms lie on opposite sides of the chain. As a result, they do not cause the chain to bend much, and their shape is similar to straight saturated fatty acids.
In most naturally occurring unsaturated fatty acids, each double bond has three (n-3), six (n-6), or nine (n-9) carbon atoms after it, and all double bonds have a cis configuration. Most fatty acids in the trans configuration (trans fats) are not found in nature and are the result of human processing (e.g., hydrogenation). Some trans fatty acids also occur naturally in the milk and meat of ruminants (such as cattle and sheep). They are produced, by fermentation, in the rumen of these animals. They are also found in dairy products from milk of ruminants, and may be also found in breast milk of women who obtained them from their diet.
The geometric differences between the various types of unsaturated fatty acids, as well as between saturated and unsaturated fatty acids, play an important role in biological processes, and in the construction of biological structures (such as cell membranes).
| Common name | Chemical structure | Δx[b] | C:D[a] | IUPAC[10] | n−x[c] |
|---|---|---|---|---|---|
| Myristoleic acid | CH3(CH2)3CH=CH(CH2)7COOH | cis-Δ9 | 14:1 | 14:1(9) | n−5 |
| Palmitoleic acid | CH3(CH2)5CH=CH(CH2)7COOH | cis-Δ9 | 16:1 | 16:1(9) | n−7 |
| Sapienic acid | CH3(CH2)8CH=CH(CH2)4COOH | cis-Δ6 | 16:1 | 16:1(6) | n−10 |
| Oleic acid | CH3(CH2)7CH=CH(CH2)7COOH | cis-Δ9 | 18:1 | 18:1(9) | n−9 |
| Elaidic acid | CH3(CH2)7CH=CH(CH2)7COOH | trans-Δ9 | 18:1 | 18:1(9t) | n−9 |
| Vaccenic acid | CH3(CH2)5CH=CH(CH2)9COOH | trans-Δ11 | 18:1 | 18:1(11t) | n−7 |
| Linoleic acid | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | cis,cis-Δ9,Δ12 | 18:2 | 18:2(9,12) | n−6 |
| Linoelaidic acid | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | trans,trans-Δ9,Δ12 | 18:2 | 18:2(9t,12t) | n−6 |
| α-Linolenic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH | cis,cis,cis-Δ9,Δ12,Δ15 | 18:3 | 18:3(9,12,15) | n−3 |
| Arachidonic acid | CH3(CH2)4CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)3COOHNIST | cis,cis,cis,cis-Δ5Δ8,Δ11,Δ14 | 20:4 | 20:4(5,8,11,14) | n−6 |
| Eicosapentaenoic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)3COOH | cis,cis,cis,cis,cis-Δ5,Δ8,Δ11,Δ14,Δ17 | 20:5 | 20:5(5,8,11,14,17) | n−3 |
| Erucic acid | CH3(CH2)7CH=CH(CH2)11COOH | cis-Δ13 | 22:1 | 22:1(13) | n−9 |
| Docosahexaenoic acid | CH3CH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCH2CH=CH(CH2)2COOH | cis,cis,cis,cis,cis,cis-Δ4,Δ7,Δ10,Δ13,Δ16,Δ19 | 22:6 | 22:6(4,7,10,13,16,19) | n−3 |
Even- vs odd-chained fatty acids
Most fatty acids are even-chained, e.g. stearic (C18) and oleic (C18), meaning they are composed of an even number of carbon atoms. Some fatty acids have odd numbers of carbon atoms; they are referred to as odd-chained fatty acids (OCFA). The most common OCFA are the saturated C15 and C17 derivatives, pentadecanoic acid and heptadecanoic acid respectively, which are found in dairy products.[11][12] On a molecular level, OCFAs are biosynthesized and metabolized slightly differently from the even-chained relatives.
Branching
Most common fatty acids are straight-chain compounds, with no additional carbon atoms bonded as side groups to the main hydrocarbon chain. Branched-chain fatty acids contain one or more methyl groups bonded to the hydrocarbon chain.
Nomenclature
Carbon atom numbering

Most naturally occurring fatty acids have an unbranched chain of carbon atoms, with a carboxyl group (–COOH) at one end, and a methyl group (–CH3) at the other end.
The position of each carbon atom in the backbone of a fatty acid is usually indicated by counting from 1 at the −COOH end. Carbon number x is often abbreviated C-x (or sometimes Cx), with x = 1, 2, 3, etc. This is the numbering scheme recommended by the IUPAC.
Another convention uses letters of the Greek alphabet in sequence, starting with the first carbon after the carboxyl group. Thus carbon α (alpha) is C-2, carbon β (beta) is C-3, and so forth.
Although fatty acids can be of diverse lengths, in this second convention the last carbon in the chain is always labelled as ω (omega), which is the last letter in the Greek alphabet. A third numbering convention counts the carbons from that end, using the labels "ω", "ω−1", "ω−2". Alternatively, the label "ω−x" is written "n−x", where the "n" is meant to represent the number of carbons in the chain.[d]
In either numbering scheme, the position of a double bond in a fatty acid chain is always specified by giving the label of the carbon closest to the carboxyl end.[d] Thus, in an 18 carbon fatty acid, a double bond between C-12 (or ω−6) and C-13 (or ω−5) is said to be "at" position C-12 or ω−6. The IUPAC naming of the acid, such as "octadec-12-enoic acid" (or the more pronounceable variant "12-octadecanoic acid") is always based on the "C" numbering.
The notation Δx,y,... is traditionally used to specify a fatty acid with double bonds at positions x,y,.... (The capital Greek letter "Δ" (delta) corresponds to Roman "D", for Double bond). Thus, for example, the 20-carbon arachidonic acid is Δ5,8,11,14, meaning that it has double bonds between carbons 5 and 6, 8 and 9, 11 and 12, and 14 and 15.
In the context of human diet and fat metabolism, unsaturated fatty acids are often classified by the position of the double bond closest between to the ω carbon (only), even in the case of multiple double bonds such as the essential fatty acids. Thus linoleic acid (18 carbons, Δ9,12), γ-linolenic acid (18-carbon, Δ6,9,12), and arachidonic acid (20-carbon, Δ5,8,11,14) are all classified as "ω−6" fatty acids; meaning that their formula ends with –CH=CH–CH
2–CH
2–CH
2–CH
2–CH
3.
Fatty acids with an odd number of carbon atoms are called odd-chain fatty acids, whereas the rest are even-chain fatty acids. The difference is relevant to gluconeogenesis.
Naming of fatty acids
The following table describes the most common systems of naming fatty acids.
| Nomenclature | Examples | Explanation |
|---|---|---|
| Trivial | Palmitoleic acid | Trivial names (or common names) are non-systematic historical names, which are the most frequent naming system used in literature. Most common fatty acids have trivial names in addition to their systematic names (see below). These names frequently do not follow any pattern, but they are concise and often unambiguous. |
| Systematic | cis-9-octadec-9-enoic acid (9Z)-octadec-9-enoic acid |
Systematic names (or IUPAC names) derive from the standard IUPAC Rules for the Nomenclature of Organic Chemistry, published in 1979,[13] along with a recommendation published specifically for lipids in 1977.[14] Carbon atom numbering begins from the carboxylic end of the molecule backbone. Double bonds are labelled with cis-/trans- notation or E-/Z- notation, where appropriate. This notation is generally more verbose than common nomenclature, but has the advantage of being more technically clear and descriptive. |
| Δx | cis-Δ9, cis-Δ12 octadecadienoic acid | In Δx (or delta-x) nomenclature, each double bond is indicated by Δx, where the double bond begins at the xth carbon–carbon bond, counting from carboxylic end of the molecule backbone. Each double bond is preceded by a cis- or trans- prefix, indicating the configuration of the molecule around the bond. For example, linoleic acid is designated "cis-Δ9, cis-Δ12 octadecadienoic acid". This nomenclature has the advantage of being less verbose than systematic nomenclature, but is no more technically clear or descriptive.[citation needed] |
| n−x (or ω−x) |
n−3 (or ω−3) |
n−x (n minus x; also ω−x or omega-x) nomenclature both provides names for individual compounds and classifies them by their likely biosynthetic properties in animals. A double bond is located on the xth carbon–carbon bond, counting from the methyl end of the molecule backbone. For example, α-linolenic acid is classified as a n−3 or omega-3 fatty acid, and so it is likely to share a biosynthetic pathway with other compounds of this type. The ω−x, omega-x, or "omega" notation is common in popular nutritional literature, but IUPAC has deprecated it in favor of n−x notation in technical documents.[13] The most commonly researched fatty acid biosynthetic pathways are n−3 and n−6. |
| Lipid numbers | 18:3 18:3n3 18:3, cis,cis,cis-Δ9,Δ12,Δ15 18:3(9,12,15) |
Lipid numbers take the form C:D,[a] where C is the number of carbon atoms in the fatty acid and D is the number of double bonds in the fatty acid. If D is more than one, the double bonds are assumed to be interrupted by CH 2 units, i.e., at intervals of 3 carbon atoms along the chain. For instance, α-linolenic acid is an 18:3 fatty acid and its three double bonds are located at positions Δ9, Δ12, and Δ15. This notation can be ambiguous, as some different fatty acids can have the same C:D numbers. Consequently, when ambiguity exists this notation is usually paired with either a Δx or n−x term.[13] For instance, although α-linolenic acid and γ-linolenic acid are both 18:3, they may be unambiguously described as 18:3n3 and 18:3n6 fatty acids, respectively. For the same purpose, IUPAC recommends using a list of double bond positions in parentheses, appended to the C:D notation.[10] For instance, IUPAC recommended notations for α- and γ-linolenic acid are 18:3(9,12,15) and 18:3(6,9,12), respectively. |
Free fatty acids
When circulating in the plasma (plasma fatty acids), not in their ester, fatty acids are known as non-esterified fatty acids (NEFAs) or free fatty acids (FFAs). FFAs are always bound to a transport protein, such as albumin.[15]
FFAs also form from triglyceride food oils and fats by hydrolysis, contributing to the characteristic rancid odor.[16] An analogous process happens in biodiesel with risk of part corrosion.
Production
Industrial
Fatty acids are usually produced industrially by the hydrolysis of triglycerides, with the removal of glycerol (see oleochemicals). Phospholipids represent another source. Some fatty acids are produced synthetically by hydrocarboxylation of alkenes.[17]
By animals
In animals, fatty acids are formed from carbohydrates predominantly in the liver, adipose tissue, and the mammary glands during lactation.[18]
Carbohydrates are converted into pyruvate by glycolysis as the first important step in the conversion of carbohydrates into fatty acids.[18] Pyruvate is then decarboxylated to form acetyl-CoA in the mitochondrion. However, this acetyl CoA needs to be transported into cytosol where the synthesis of fatty acids occurs. This cannot occur directly. To obtain cytosolic acetyl-CoA, citrate (produced by the condensation of acetyl-CoA with oxaloacetate) is removed from the citric acid cycle and carried across the inner mitochondrial membrane into the cytosol.[18] There it is cleaved by ATP citrate lyase into acetyl-CoA and oxaloacetate. The oxaloacetate is returned to the mitochondrion as malate.[19] The cytosolic acetyl-CoA is carboxylated by acetyl-CoA carboxylase into malonyl-CoA, the first committed step in the synthesis of fatty acids.[19][20]
Malonyl-CoA is then involved in a repeating series of reactions that lengthens the growing fatty acid chain by two carbons at a time. Almost all natural fatty acids, therefore, have even numbers of carbon atoms. When synthesis is complete the free fatty acids are nearly always combined with glycerol (three fatty acids to one glycerol molecule) to form triglycerides, the main storage form of fatty acids, and thus of energy in animals. However, fatty acids are also important components of the phospholipids that form the phospholipid bilayers out of which all the membranes of the cell are constructed (the cell wall, and the membranes that enclose all the organelles within the cells, such as the nucleus, the mitochondria, endoplasmic reticulum, and the Golgi apparatus).[18]
The "uncombined fatty acids" or "free fatty acids" found in the circulation of animals come from the breakdown (or lipolysis) of stored triglycerides.[18][21] Because they are insoluble in water, these fatty acids are transported bound to plasma albumin. The levels of "free fatty acids" in the blood are limited by the availability of albumin binding sites. They can be taken up from the blood by all cells that have mitochondria (with the exception of the cells of the central nervous system). Fatty acids can only be broken down in mitochondria, by means of beta-oxidation followed by further combustion in the citric acid cycle to CO2 and water. Cells in the central nervous system, although they possess mitochondria, cannot take free fatty acids up from the blood, as the blood–brain barrier is impervious to most free fatty acids,[citation needed] excluding short-chain fatty acids and medium-chain fatty acids.[22][23] These cells have to manufacture their own fatty acids from carbohydrates, as described above, in order to produce and maintain the phospholipids of their cell membranes, and those of their organelles.[18]
Variation between animal species
Studies on the cell membranes of mammals and reptiles discovered that mammalian cell membranes are composed of a higher proportion of polyunsaturated fatty acids (DHA, omega-3 fatty acid) than reptiles.[24] Studies on bird fatty acid composition have noted similar proportions to mammals but with 1/3rd less omega-3 fatty acids as compared to omega-6 for a given body size.[25] This fatty acid composition results in a more fluid cell membrane but also one that is permeable to various ions (H+ & Na+), resulting in cell membranes that are more costly to maintain. This maintenance cost has been argued to be one of the key causes for the high metabolic rates and concomitant warm-bloodedness of mammals and birds.[24] However polyunsaturation of cell membranes may also occur in response to chronic cold temperatures as well. In fish increasingly cold environments lead to increasingly high cell membrane content of both monounsaturated and polyunsaturated fatty acids, to maintain greater membrane fluidity (and functionality) at the lower temperatures.[26][27]
Fatty acids in dietary fats
The following table gives the fatty acid, vitamin E and cholesterol composition of some common dietary fats.[28][29]
| Saturated | Monounsaturated | Polyunsaturated | Cholesterol | Vitamin E | |
|---|---|---|---|---|---|
| g/100g | g/100g | g/100g | mg/100g | mg/100g | |
| Animal fats | |||||
| Duck fat[30] | 33.2 | 49.3 | 12.9 | 100 | 2.70 |
| Lard[30] | 40.8 | 43.8 | 9.6 | 93 | 0.60 |
| Tallow[30] | 49.8 | 41.8 | 4.0 | 109 | 2.70 |
| Butter | 54.0 | 19.8 | 2.6 | 230 | 2.00 |
| Vegetable fats | |||||
| Coconut oil | 85.2 | 6.6 | 1.7 | 0 | .66 |
| Cocoa butter | 60.0 | 32.9 | 3.0 | 0 | 1.8 |
| Palm kernel oil | 81.5 | 11.4 | 1.6 | 0 | 3.80 |
| Palm oil | 45.3 | 41.6 | 8.3 | 0 | 33.12 |
| Cottonseed oil | 25.5 | 21.3 | 48.1 | 0 | 42.77 |
| Wheat germ oil | 18.8 | 15.9 | 60.7 | 0 | 136.65 |
| Soybean oil | 14.5 | 23.2 | 56.5 | 0 | 16.29 |
| Olive oil | 14.0 | 69.7 | 11.2 | 0 | 5.10 |
| Corn oil | 12.7 | 24.7 | 57.8 | 0 | Zdroj:https://en.wikipedia.org?pojem=Long_chain_fatty_acids|
