
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hydrogen peroxide
| |||
| Systematic IUPAC name
Peroxol | |||
| Other names
Dioxidane
Oxidanyl Perhydroxic acid 0-hydroxyol Oxygenated water Peroxaan | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.028.878 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2015 (>60% soln.) 2014 (20–60% soln.) 2984 (8–20% soln.) | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| H2O2 | |||
| Molar mass | 34.014 g·mol−1 | ||
| Appearance | Very light blue liquid | ||
| Odor | slightly sharp | ||
| Density | 1.11 g/cm3 (20 °C, 30% (w/w) solution)[1] 1.450 g/cm3 (20 °C, pure) | ||
| Melting point | −0.43 °C (31.23 °F; 272.72 K) | ||
| Boiling point | 150.2 °C (302.4 °F; 423.3 K) (decomposes) | ||
| Miscible | |||
| Solubility | soluble in ether, alcohol insoluble in petroleum ether | ||
| log P | −0.43[2] | ||
| Vapor pressure | 5 mmHg (30 °C)[3] | ||
| Acidity (pKa) | 11.75 | ||
| −17.7·10−6 cm3/mol | |||
Refractive index (nD)
|
1.4061 | ||
| Viscosity | 1.245 cP (20 °C) | ||
| 2.26 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
1.267 J/(g·K) (gas) 2.619 J/(g·K) (liquid) | ||
Std enthalpy of
formation (ΔfH⦵298) |
−187.80 kJ/mol | ||
| Pharmacology | |||
| A01AB02 (WHO) D08AX01 (WHO), D11AX25 (WHO), S02AA06 (WHO) | |||
| Hazards | |||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H271, H302, H314, H332, H335, H412 | |||
| P280, P305+P351+P338, P310 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
1518 mg/kg[citation needed] 2000 mg/kg (oral, mouse)[4] | ||
LC50 (median concentration)
|
1418 ppm (rat, 4 hr)[4] | ||
LCLo (lowest published)
|
227 ppm (mouse)[4] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 1 ppm (1.4 mg/m3)[3] | ||
REL (Recommended)
|
TWA 1 ppm (1.4 mg/m3)[3] | ||
IDLH (Immediate danger)
|
75 ppm[3] | ||
| Safety data sheet (SDS) | ICSC 0164 (>60% soln.) | ||
| Related compounds | |||
Related compounds
|
Water Ozone Hydrazine Hydrogen disulfide Dioxygen difluoride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
Hydrogen peroxide is a chemical compound with the formula H2O2. In its pure form, it is a very pale blue[5] liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or "high-test peroxide", decomposes explosively when heated and has been used as both a monopropellant and an oxidizer in rocketry.[6]
Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly into water and elemental oxygen when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a stabilizer in a weakly acidic solution in an opaque bottle. Hydrogen peroxide is found in biological systems including the human body. Enzymes that use or decompose hydrogen peroxide are classified as peroxidases.
Properties
The boiling point of H2O2 has been extrapolated as being 150.2 °C (302.4 °F), approximately 50 °C (90 °F) higher than water. In practice, hydrogen peroxide will undergo potentially explosive thermal decomposition if heated to this temperature. It may be safely distilled at lower temperatures under reduced pressure.[7]
Hydrogen peroxide forms stable adducts with urea (Hydrogen peroxide - urea), sodium carbonate (sodium percarbonate) and other compounds.[8] An acid-base adduct with triphenylphosphine oxide is a useful "carrier" for H2O2 in some reactions.
Structure
Hydrogen peroxide (H2O2) is a nonplanar molecule with (twisted) C2 symmetry; this was first shown by Paul-Antoine Giguère in 1950 using infrared spectroscopy.[9][10] Although the O−O bond is a single bond, the molecule has a relatively high rotational barrier of 386 cm−1 (4.62 kJ/mol) for rotation between enantiomers via the trans configuration, and 2460 cm−1 (29.4 kJ/mol) via the cis configuration.[11] These barriers are proposed to be due to repulsion between the lone pairs of the adjacent oxygen atoms and dipolar effects between the two O–H bonds. For comparison, the rotational barrier for ethane is 1040 cm−1 (12.4 kJ/mol).
The approximately 100° dihedral angle between the two O–H bonds makes the molecule chiral. It is the smallest and simplest molecule to exhibit enantiomerism. It has been proposed that the enantiospecific interactions of one rather than the other may have led to amplification of one enantiomeric form of ribonucleic acids and therefore an origin of homochirality in an RNA world.[12]
The molecular structures of gaseous and crystalline H2O2 are significantly different. This difference is attributed to the effects of hydrogen bonding, which is absent in the gaseous state.[13] Crystals of H2O2 are tetragonal with the space group D4
4 or P41212.[14]
Aqueous solutions
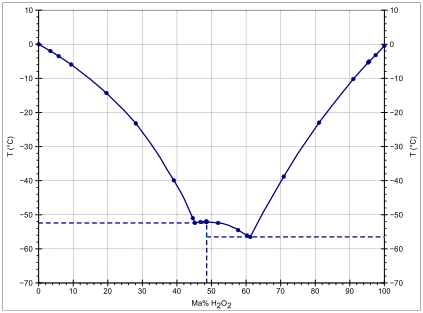
In aqueous solutions, hydrogen peroxide forms a eutectic mixture, exhibiting freezing-point depression down as low as -56 °C; pure water has a freezing point of 0 °C and pure hydrogen peroxide of -0.43 °C. The boiling point of the same mixtures is also depressed in relation with the mean of both boiling points (125.1 °C). It occurs at 114 °C. This boiling point is 14 °C greater than that of pure water and 36.2 °C less than that of pure hydrogen peroxide.[15]
| H2O2 (w/w) | Density (g/cm3) |
Temp. (°C) |
|---|---|---|
| 3% | 1.0095 | 15 |
| 27% | 1.10 | 20 |
| 35% | 1.13 | 20 |
| 50% | 1.20 | 20 |
| 70% | 1.29 | 20 |
| 75% | 1.33 | 20 |
| 96% | 1.42 | 20 |
| 98% | 1.43 | 20 |
| 100% | 1.45 | 20 |
Hydrogen peroxide is most commonly available as a solution in water. For consumers, it is usually available from pharmacies at 3 and 6 wt% concentrations. The concentrations are sometimes described in terms of the volume of oxygen gas generated; one milliliter of a 20-volume solution generates twenty milliliters of oxygen gas when completely decomposed. For laboratory use, 30 wt% solutions are most common. Commercial grades from 70% to 98% are also available, but due to the potential of solutions of more than 68% hydrogen peroxide to be converted entirely to steam and oxygen (with the temperature of the steam increasing as the concentration increases above 68%) these grades are potentially far more hazardous and require special care in dedicated storage areas. Buyers must typically allow inspection by commercial manufacturers.
Comparison with analogues
Zdroj:https://en.wikipedia.org?pojem=Hydrogen_peroxideText je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk





