
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |

| |
| Names | |
|---|---|
| IUPAC name
Triscobalt (III) ion
| |
| Identifiers | |
3D model (JSmol)
|
|
| |
| |
| Properties | |
| Co4H42N12O18S3 | |
| Molar mass | 830.31 g·mol−1 |
| Sparingly soluble in water [1] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C 77 °F, 100 kPa).
| |
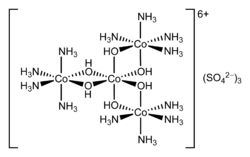
In chemistry, hexol is a cation with formula {Co(NH3)4(OH)23Co}6+ — a coordination complex consisting of four cobalt cations in oxidation state +3, twelve ammonia molecules NH
3, and six hydroxy anions HO−
, with a net charge of +6. The hydroxy groups act as bridges between the central cobalt atom and the other three, which carry the ammonia ligands.
Salts of hexol, such as the sulfate {Co(NH3)4(OH)23Co}(SO4)3(H2O)x, are of historical significance as the first synthetic non-carbon-containing chiral compounds.[2] [3]
Preparationedit
Salts of hexol were first described by Jørgensen,[4] although it was Werner who recognized its structure.[5] The cation is prepared by heating a solution containing the cis-diaquotetramminecobalt(III) cation Co(NH3)4(H2O)23+ with a dilute base:[1]
- 4 Co(NH3)4(H2O)23+ + 2 HO− → {Co(NH3)4(OH)23Co}6+ + 4 NH4+ + 4 H2O
Hexol sulfateedit
Starting with the sulfate and using ammonium hydroxide as the base, depending on the conditions, one obtains the 9-hydrate, the 6-hydrate, or the 4-hydrate of hexol sulfate. These salts form dark brownish-violet or black tabular crystals, with low solubility in water. When treated with concentrated hydrochloric acid, hexol sulfate converts to cis-diaquotetramminecobalt(III) sulfate. In boiling dilute sulfuric acid, hexol sulfate further degrades with evolution of oxygen and nitrogen.[1]
Optical propertiesedit
The hexol cation exists as two optical isomers that are mirror images of each other, depending on the arrangement of the bonds between the central cobalt atom and the three bidentate peripheral units Co(NH3)4(HO)2. It belongs to the D3 point group. The nature of chirality can be compared to that of the ferrioxalate anion Fe(C
2O
4)
33−
.
In a historic set of experiments, a salt of hexol with an optically active anion — specifically, its D-(+)-bromocamphorsulfonate – was resolved into separate salts of the two cation isomers by fractional crystallisation.[5] A more efficient resolution involves the bis(tartrato)diantimonate(III) anion. The hexol hexacation has a high specific rotation of 2640°.[6]
"Second hexol"edit
Werner also described a second achiral hexol (a minor byproduct from the production of Fremy's salt) that he incorrectly identified as a linear tetramer. The second hexol is hexanuclear (contains six cobalt centres in each ion), not tetranuclear.[7] Its point group is C2h, and its formula is Co
6(NH
3)
14(OH)
8O
26+
, whereas that of hexol is Co
4(NH
3)
12(OH)
66+
.
Referencesedit
- ^ a b c Kauffman, George B.; Pinnell, Robert P. (1960). "Tris[tetrammine-μ-dihydroxo-cobalt(III)]cobalt(III) Sulfate 4-Hydrate". TrisTetrammine-μ-Dihydroxo-Cobalt(III) Cobalt(III) Sulfate 4-Hydrate. Inorganic Syntheses. Vol. 6. pp. 176–179. doi:10.1002/9780470132371.ch56. ISBN 9780470132371.
- ^ Miessler, G. L. and Tarr, D. A. Inorganic Chemistry, 3rd ed., Pearson/Prentice Hall publisher, ISBN 0-13-035471-6.
- ^ Ernst, Karl-Heinz; Berke, Heinz (2011). "Optical Activity and Alfred Werner's Coordination Chemistry". Chirality. 23 (3): 187–189. doi:10.1002/chir.20912. PMID 20928897.
- ^ Jørgensen, S. M. (1898). "Zur Konstitution der Kobalt-, Chrom- und Rhodiumbasen". Zeitschrift für Anorganische Chemie. 16: 184–197. doi:10.1002/zaac.18980160116.
- ^ a b Werner, A. (1907). "Über mehrkernige Metallammoniake" [Poly-nucleated Metal-amines]. Ber. Dtsch. Chem. Ges. (in German). 40 (2): 2103–2125. doi:10.1002/cber.190704002126.
- ^ Yasui, Takaji; Ama, Tomoharu; Kauffman, George B. (1992). "Resolution of the Dodecaamminehexa-μ-Hydroxo-Tetracobalt(III) Ion". Inorganic Syntheses. Vol. 29. pp. 169–174. doi:10.1002/9780470132609.ch41. ISBN 9780470132609.
- ^ Jackson, W. Gregory; McKeon, Josephine A.; Zehnder, Margareta; Neuberger, Markus; Fallab, Silvio (2004). "The rediscovery of Alfred Werner's second hexol". Chemical Communications (20): 2322–2323. doi:10.1039/B408277J. PMID 15490001.
External linksedit
- Hexol Molecule of the Month September 1997 Website
- National Pollutant Inventory – Cobalt fact sheet
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk

