
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |

| |
| Names | |
|---|---|
| IUPAC name
Chromium(III) acetate hydrate
| |
| Other names
chromic acetate,
chromium triacetate, chromium(III) ethanoate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.012.646 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H36ClCr3O22 | |
| Molar mass | 723.84 g·mol−1 |
| Appearance | grayish-green to blueish-green solid |
| Density | 1.662 g/cm3 |
| Melting point | 1,152[1] °C (2,106 °F; 1,425 K) |
| -5104.0·10−6 cm3/mol | |
| Structure | |
| octahedral | |
| Related compounds | |
Related compounds
|
Manganese(III) acetate Iron(III) acetate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
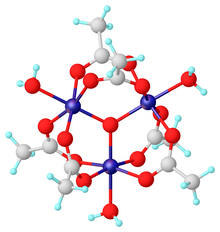
Chromium(III) acetate, commonly known as basic chromium acetate,[2] describes a family of salts where the cation has the formula +. The trichromium cation is encountered with a variety of anions, such as chloride and nitrate. Data in the table above are for the chloride hexahydrate, Cl(H2O)6.
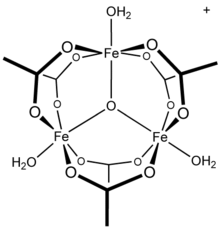
Salts of basic chromium acetate has long attracted interest because of its distinctive structure, which features octahedral Cr(III) centers, a triply bridging oxo ligand, six acetate ligands, and three aquo ligands.[2] The same structure is shared with basic iron acetate and basic manganese acetate.[2][3] Little evidence exists for a simple chromium(III) acetate, i.e. lacking the oxo ligand.[4] Chromium(III) acetate is a blue/grey-green powder, which is soluble in water. It is still[3] prepared according to the original procedure from 1909.[5]
See also
References
- ^ "Chromium (III) compounds". National Pollutant Inventory. Australian Government Department of Agriculture, Water and the Environment. Retrieved 14 April 2021.
- ^ a b c Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ a b Kurt J. Schenk; Hans U. Güdel (1982). "Low-temperature structural and spectroscopic properties of Cl.6H2O". Inorg. Chem. 21 (6): 2253–2256. doi:10.1021/ic00136a025.
- ^ Erre, Liliana Strinna; Micera, Giovanni; Glowiak, Tadeusz; Kozlowski, Henry (April 1997). "Chromium (III) Acetate, Chromium (III) Acetate Hydroxide, or µ3-Oxo-esakis-(µ2-acetato-O,O') - triaqua-trichromium (III) Acetate? Determining the Structure of a Complex Compound by Analytical and Spectroscopic Methods". Journal of Chemical Education. 74 (4): 432. Bibcode:1997JChEd..74..432E. doi:10.1021/ed074p432.
- ^ R. Weinland; P. Dinkelacker (1909). "Über Salze einer Hexaacetato(formiato)‐trichrombase. II". Berichte der Deutschen Chemischen Gesellschaft. 42 (3): 2997–3018. doi:10.1002/cber.19090420318.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
