
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
3,4-Dihydro-2H-naphthalen-1-one | |
| Other names
α-Tetralone; 1-Tetralone
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.692 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H10O | |
| Molar mass | 146.189 g·mol−1 |
| Appearance | colorless liquid |
| Density | 1.099 g·cm−3 (25 °C)[1] |
| Melting point | 2–7 °C[1] |
| Boiling point | 255–257 °C[2] 113–116 °C (8 hPa)[1] |
| insoluble[3] | |
| Solubility | soluble in organic solvents |
| Vapor pressure | 2.7 Pa (20 °C)[3] |
Refractive index (nD)
|
1.5672 |
| Hazards | |
| GHS labelling:[4] | |

| |
| Warning | |
| H302 | |
| P264, P270, P301+P317, P330, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
1-Tetralone is a bicyclic aromatic hydrocarbon and a ketone. In terms of its structure, it can also be regarded as benzo-fused cyclohexanone. It is a colorless oil with a faint odor.[5] It is used as starting material for agricultural and pharmaceutical agents. The carbon skeleton of 1-tetralone is found in natural products such as Aristelegone A (4,7-dimethyl-6-methoxy-1-tetralone) from the family of Aristolochiaceae used in traditional Chinese medicine.[6]
Preparation
By oxidation of 1,2,3,4-tetrahydronaphthalene
As already described in 1933 by Heinrich Hock, 1,2,3,4-tetrahydronaphthalene tends to autoxidize and gradually forms the 1-hydroperoxide with atmospheric oxygen.[7] The heavy metal ion catalyzed air oxidation of 1,2,3,4-tetrahydronaphthalene with Cr3+[8] or Cu2+ in the liquid phase leads via the hydroperoxide to a mixture of the intermediate 1-tetralol and the final product 1-tetralone.[9]

The boiling points of the main component 1-tetralone (255-257 °C) and the minor component 1-tetralol (255 °C)[2] are virtually identical, the latter is therefore removed by a chemical reaction.[10]
By Friedel-Crafts reactions
The starting compound 4-phenylbutanoic acid is accessible from 3-benzoylpropanoic acid via catalytic hydrogenation, using a palladium contact catalyst.[5] 3-Benzoylpropanoic acid[11] itself can be obtained by a Haworth reaction (a variant of the Friedel-Crafts reaction) from benzene and succinic anhydride.
The intramolecular cyclization of 4-phenylbutanoic acid to 1-tetralone is catalyzed by polyphosphoric acid[5] and methanesulfonic acid.[12]

It has been described as a teaching experiment for chemistry lessons.[13] 4-Phenylbutanoic acid can also be quantitatively converted into 1-tetralone by heating in the presence of a strong Lewis acid catalyst such as bismuth(III)bis(trifluoromethanesulfonyl)amide[14] , which is relatively easily accessible.[15]
The use of the acid chloride and tin(IV) chloride (SnCl4) allows significantly shorter reaction times than the Friedel-Crafts acylation with 4-phenylbutanoic acid.[10]

4-Phenylbutanoic acid chlorides with electron-donating groups can be cyclized to 1-tetralones under mild reaction conditions in yields greater than 90% using the strong hydrogen-bonding solvent hexafluoroisopropanol (HFIP).[16]
The AlCl3-catalyzed acylation of benzene with γ-butyrolactone produces 1-tetralone.[10]

Reactions
1-Tetralone can be reduced via a Birch reduction with lithium in liquid ammonia to 1,2,3,4-tetrahydronaphthalene.[17] The keto group can also be reduced to a secondary alcohol giving 1-tetralol, when a modified process is applied, using the addition of aqueous ammonium chloride solution after evaporation of the ammonia.[18]

With calcium in liquid ammonia, 1-tetralone is reduced to 1-tetralol at -33 °C in 81% yield.[19]
The methylene group in α-position to the keto group is particularly reactive and can be converted with formaldehyde (in the form of the trimeric trioxane) to 2-methylene-1-tetralone in the presence of the trifluoroacetic acid salt of N-methylaniline with yields up to 91% .
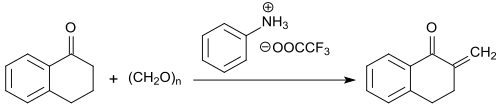
The 2-methylene ketone is stable at temperatures below -5 °C, but fully polymerizes at room temperature within 12 hours.[20]
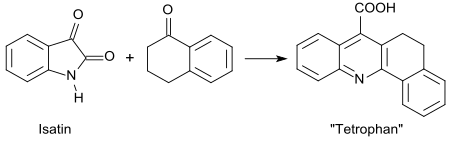
In the Pfitzinger reaction of 1-tetralone with isatin, a compound called tetrofan (3,4-dihydro-1,2-benzacridine-5-carboxylic acid) is formed.
The reactivity of the α-methylene group is also exploited in the reaction of 1-tetralone with methanol at 270-290 °C, which produces via dehydrogenation and formation of the aromatic naphthalene ring system 2-methyl-1-naphthol in 66% yield.[21]

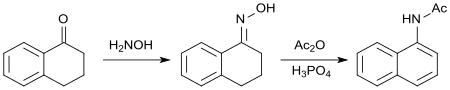
The oxime of 1-tetralone reacts with acetic anhydride leading to aromatization of the cycloalkanone ring. The resulting N-(1-naphthyl)acetamide[22] has biological properties akin to those of 2-(1-Naphthyl)acetic acid as a synthetic auxin.
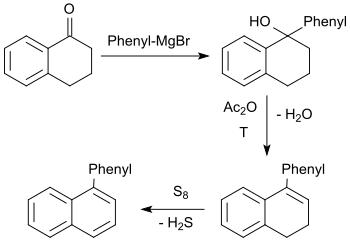
The tertiary alcohol formed in the Grignard reaction of 1-tetralone with phenylmagnesium bromide reacts with acetic anhydride upon elimination of water to 1-phenyl-3,4-dihydronaphthalene, which is dehydrated with elemental sulfur in an overall yield of about 45% to 1-phenylnaphthalene.[23]
The ruthenium(II)-catalyzed arylation of 1-tetralone using phenyl boronic acid neopentyl glycol ester gives 8-phenyl-1-tetralone in up to 86% yield.[24]

With 5-aminotetrazole and an aromatic aldehyde, 1-tetralone reacts in a multi-component reaction under microwave irradiation to form a four-membered heterocyclic ring system.[25]

Applications
By far the most important application of 1-tetralone is in the synthesis of 1-naphthol by aromatization, e.g. upon contact with platinum catalysts at 200 to 450 °C.[26]
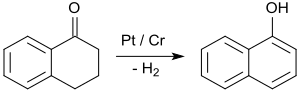
1-Naphthol is the starting material for the insecticides carbaryl and the beta-blockers propranolol.
Safety
Toxicological studies were dermally performed with rabbits, with an LD50 of 2192 mg·kg−1 body weight being observed.[1]
References
- ^ a b c d Sigma-Aldrich Co., α-Tetralon. Retrieved on 25. November 2017.
- ^ a b William M. Haynes (2016), CRC Handbook of Chemistry and Physics, 97th Edition, Boca Raton, FL, U.S.A.: CRC Press, pp. 3–504, ISBN 978-1-4987-5429-3
- ^ a b "alpha-Tetralone 529-34-0 | TCI Deutschland GmbH". www.tcichemicals.com (in German). Retrieved 2017-12-17.
- ^ "1-Tetralone". pubchem.ncbi.nlm.nih.gov.
- ^ a b c H.R. Snyder, F.X. Werber (1940). "α-Tetralone". Org. Synth. 20: 94. doi:10.15227/orgsyn.020.0094.
- ^ P.-C. Kuo; Y.-C. Li; T.-S. Wu (2012), "Chemical constituents and pharmacology of the Aristolochia species", EJTCM, vol. 2, no. 4, pp. 249–266, doi:10.1016/S2225-4110(16)30111-0, PMC 3942903, PMID 24716140
- ^ H. Hock; W. Susemihl (1933), "Autoxydation von Kohlenwasserstoffen: Über ein durch Autoxydation erhaltenes Tetrahydro-naphthalin-peroxyd (I. Mitteil.)", Ber. Dtsch. Chem. Ges. (in German), vol. 66, no. 1, pp. 61–68, doi:10.1002/cber.19330660113
- ^ S. Bhattacharjee; Y.-R. Lee; W.-S. Ahn (2017), "Oxidation of tetraline to 1-tetralone over CrAPO-5", Korean J. Chem. Eng. (in German), vol. 34, no. 3, pp. 701–705, doi:10.1007/s11814-016-0310-4, S2CID 100124813
- ^ US 4473711, R.W. Coon, "Liquid-phase process for oxidation of tetralin", published 1984-09-25, assigned to Union Carbide Corp.
- ^ a b c C.E. Olson, A.R. Bader (1955). "α-Tetralone". Org. Synth. 35: 95. doi:10.15227/orgsyn.035.0095.
- ^ L. F. Somerville, C. F. H. Allen (1933). "β-Benzoylpropionic acid". Org. Synth. 13: 12. doi:10.15227/orgsyn.013.0012.
- ^ V. Premasagar; V.A. Palaniswamy; E.J. Eisenbraun (1981), "Methanesulfonic acid catalyzed cyclization of 3-arylpropanoic and 4-arylbutanoic acids to 1-indanones and 1-tetralones", J. Org. Chem., vol. 46, no. 14, pp. 2974–2976, doi:10.1021/jo00325a028
- ^ M.S. Holden; R.D. Crouch; K.A. Barker (2005), "Formation of α-tetralone by intramolecular Friedel-Crafts acylation", J. Chem. Educ., vol. 82, no. 6, pp. 934–935, Bibcode:2005JChEd..82..934H, doi:10.1021/ed082p934
- ^ S. Antoniotti; E. Dunach (2008), "Facile preparation of metallic triflates and triflimidates by oxidative dissolution of metal powders", Chem. Commun., vol. 8, no. 8, pp. 993–995, doi:10.1039/B717689A, PMID 18283360
- ^ D.-M. Cui; M. Kawamura; S. Shimada; T. Hayashi; M. Tanaka (2003), "Synthesis of 1-tetralones by intramolecular Friedel-Crafts reaction of 4-arylbutyric acids using Lewis acid catalysts", Tetrahedron Lett., vol. 44, no. 21, pp. 4007–4010, doi:10.1016/S0040-4039(03)00855-4
- ^ H. Motiwala; R.H. Vekariya; J. Aubé (2015), "Intramolecular Friedel-Crafts acylation reaction promoted by 1,1,1,3,3,3-hexafluoro-2-propanol", Org. Lett. (in German), vol. 17, no. 21, pp. 5484–5487, doi:10.1021/acs.orglett.5b02851, PMID 26496158
- ^ S.S. Hall; S.D. Lipsky; F.J. McEnroe; A.P. Bartels (1971), "Lithium-ammonia Reduction of Aromatic Ketones to Aromatic Hydrocarbons", J. Org. Chem., vol. 38, no. 18, pp. 2588–2591, doi:10.1021/jo00817a004
- ^ Z. Marcinow; P.W. Rabideau (1988), "Metal-Ammonia Reduction of α-Tetralone. Competition Between Ring Reduction, Carbonyl Reduction, and Dimer Formation", J. Org. Chem., vol. 53, no. 9, pp. 2117–2119, doi:10.1021/jo00244a054
- ^ J.R. Hwu; Y.S. Wein; Y.-J. Leu (1996), "Calcium metal in liquid ammonia for selective reduction of organic compounds", J. Org. Chem. (in German), vol. 61, no. 4, pp. 1493–1499, doi:10.1021/jo951219c
- ^ "Methylene ketones and aldehydes by simple, direct methylene transfer: 2-Methylene-1-oxo-1,2,3,4-tetrahydronaphthalene". Organic Syntheses. doi:10.15227/orgsyn.060.0088.
- ^ I. Yuranov; L. Kiwi-Minsker; A. Renken (2002), "One-step vapour-phase synthesis of 2-methyl-1-naphthol from 1-tetralone", Appl. Catal. A (in German), vol. 226, no. 1–2, pp. 193–198, doi:10.1016/S0926-860X(01)00902-4
- ^ M.S. Newman; W.M. Hung (1973), "An improved aromatization of α-tetralone oximes to N-(1-naphthyl)acetamides", J. Org. Chem. (in German), vol. 38, no. 23, pp. 4073–4074, doi:10.1021/jo00987a029
- ^ "1-Phenylnaphthalene". Organic Syntheses. doi:10.15227/orgsyn.024.0084.
- ^ "Ruthenium-catalyzed arylation of ortho C-H bond in an aromatic with an arylboronate: 8-Phenyl-1-tetralone". Organic Syntheses. doi:10.15227/orgsyn.087.0209.
- ^ G.P. Kantin; M. Krasavin (2016), "Reaction of α-tetralone, 1H-tetrazol-5-amine, and aromatic aldehydes upon microwave irradiation – a convenient method for the synthesis of 5,6,7,12-tetrahydrobenzotetrazoloquinazolines", Chem. Heterocycl. Compd. (in German), vol. 52, no. 11, pp. 918–922, doi:10.1007/s10593-017-1985-0, S2CID 99144245
- ^ DE 2421745, K. Kudo, T. Ohmae, A. Uno, "Verfahren zur Herstellung von α-Naphthol durch katalytische Dehydrierung von α-Tetralon", published 1975-11-20, assigned to Sumitomo Chemical Co., Ltd.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
