
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| Planck constant | |
|---|---|
Common symbols | |
| SI unit | joule per hertz (joule seconds) |
Other units | electronvolt per hertz (electronvolt seconds) |
| In SI base units | kg m2 s−1 |
| Dimension | |
| Value | 6.62607015×10−34 J⋅Hz−1 4.135667696...×10−15 eV⋅Hz−1 |
| Reduced Planck constant | |
|---|---|
Common symbols | |
| SI unit | joule-seconds |
Other units | electronvolt-seconds |
| In SI base units | kg m2 s−1 |
Derivations from other quantities | |
| Dimension | |
| Value | 1.054571817...×10−34 J⋅s 6.582119569...×10−16 eV⋅s |
The Planck constant, or Planck's constant, denoted by ,[1] is a fundamental physical constant[1] of foundational importance in quantum mechanics: a photon's energy is equal to its frequency multiplied by the Planck constant, and the wavelength of a matter wave equals the Planck constant divided by the associated particle momentum.
The constant was postulated by Max Planck in 1900 as a proportionality constant needed to explain experimental black-body radiation.[2] Planck later referred to the constant as the "quantum of action".[3] In 1905, Albert Einstein associated the "quantum" or minimal element of the energy to the electromagnetic wave itself. Max Planck received the 1918 Nobel Prize in Physics "in recognition of the services he rendered to the advancement of Physics by his discovery of energy quanta".
In metrology, the Planck constant is used, together with other constants, to define the kilogram, the SI unit of mass.[4] The SI units are defined in such a way that, when the Planck constant is expressed in SI units, it has the exact value = 6.62607015×10−34 J⋅Hz−1.[5][6]
History
Origin of the constant

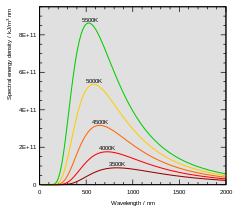
Planck's constant was formulated as part of Max Planck's successful effort to produce a mathematical expression that accurately predicted the observed spectral distribution of thermal radiation from a closed furnace (black-body radiation).[7] This mathematical expression is now known as Planck's law.
In the last years of the 19th century, Max Planck was investigating the problem of black-body radiation first posed by Kirchhoff some 40 years earlier. Every physical body spontaneously and continuously emits electromagnetic radiation. There was no expression or explanation for the overall shape of the observed emission spectrum. At the time, Wien's law fit the data for short wavelengths and high temperatures, but failed for long wavelengths.[7]: 141 Also around this time, but unknown to Planck, Lord Rayleigh had derived theoretically a formula, now known as the Rayleigh–Jeans law, that could reasonably predict long wavelengths but failed dramatically at short wavelengths.
Approaching this problem, Planck hypothesized that the equations of motion for light describe a set of harmonic oscillators, one for each possible frequency. He examined how the entropy of the oscillators varied with the temperature of the body, trying to match Wien's law, and was able to derive an approximate mathematical function for the black-body spectrum,[2] which gave a simple empirical formula for long wavelengths.
Planck tried to find a mathematical expression that could reproduce Wien's law (for short wavelengths) and the empirical formula (for long wavelengths). This expression included a constant, , which is thought to be for Hilfsgrösse (auxiliary variable),[8] and subsequently became known as the Planck constant. The expression formulated by Planck showed that the spectral radiance of a body for frequency ν at absolute temperature T is given by
- ,
where is the Boltzmann constant, is the Planck constant, and is the speed of light in the medium, whether material or vacuum.[9][10][11]
The spectral radiance of a body, , describes the amount of energy it emits at different radiation frequencies. It is the power emitted per unit area of the body, per unit solid angle of emission, per unit frequency. The spectral radiance can also be expressed per unit wavelength instead of per unit frequency. In this case, it is given by
- ,
showing how radiated energy emitted at shorter wavelengths increases more rapidly with temperature than energy emitted at longer wavelengths.[12]
Planck's law may also be expressed in other terms, such as the number of photons emitted at a certain wavelength, or the energy density in a volume of radiation. The SI units of are W·sr−1·m−2·Hz−1, while those of are W·sr−1·m−3.
Planck soon realized that his solution was not unique. There were several different solutions, each of which gave a different value for the entropy of the oscillators.[2] To save his theory, Planck resorted to using the then-controversial theory of statistical mechanics,[2] which he described as "an act of desperation".[13] One of his new boundary conditions was
to interpret UN not as a continuous, infinitely divisible quantity, but as a discrete quantity composed of an integral number of finite equal parts. Let us call each such part the energy element ε;
— Planck, On the Law of Distribution of Energy in the Normal Spectrum[2]
With this new condition, Planck had imposed the quantization of the energy of the oscillators, "a purely formal assumption ... actually I did not think much about it ..." in his own words,[14] but one that would revolutionize physics. Applying this new approach to Wien's displacement law showed that the "energy element" must be proportional to the frequency of the oscillator, the first version of what is now sometimes termed the "Planck–Einstein relation":
Planck was able to calculate the value of from experimental data on black-body radiation: his result, 6.55×10−34 J⋅s, is within 1.2% of the currently defined value.[2] He also made the first determination of the Boltzmann constant from the same data and theory.[15]
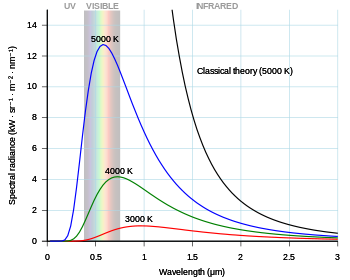
Development and application
The black-body problem was revisited in 1905, when Lord Rayleigh and James Jeans (on the one hand) and Albert Einstein (on the other hand) independently proved that classical electromagnetism could never account for the observed spectrum. These proofs are commonly known as the "ultraviolet catastrophe", a name coined by Paul Ehrenfest in 1911. They contributed greatly (along with Einstein's work on the photoelectric effect) in convincing physicists that Planck's postulate of quantized energy levels was more than a mere mathematical formalism. The first Solvay Conference in 1911 was devoted to "the theory of radiation and quanta".[16]
Photoelectric effect
The photoelectric effect is the emission of electrons (called "photoelectrons") from a surface when light is shone on it. It was first observed by Alexandre Edmond Becquerel in 1839, although credit is usually reserved for Heinrich Hertz,[17] who published the first thorough investigation in 1887. Another particularly thorough investigation was published by Philipp Lenard (Lénárd Fülöp) in 1902.[18] Einstein's 1905 paper[19] discussing the effect in terms of light quanta would earn him the Nobel Prize in 1921,[17] after his predictions had been confirmed by the experimental work of Robert Andrews Millikan.[20] The Nobel committee awarded the prize for his work on the photo-electric effect, rather than relativity, both because of a bias against purely theoretical physics not grounded in discovery or experiment, and dissent amongst its members as to the actual proof that relativity was real.[21][22]
Before Einstein's paper, electromagnetic radiation such as visible light was considered to behave as a wave: hence the use of the terms "frequency" and "wavelength" to characterize different types of radiation. The energy transferred by a wave in a given time is called its intensity. The light from a theatre spotlight is more intense than the light from a domestic lightbulb; that is to say that the spotlight gives out more energy per unit time and per unit space (and hence consumes more electricity) than the ordinary bulb, even though the color of the light might be very similar. Other waves, such as sound or the waves crashing against a seafront, also have their intensity. However, the energy account of the photoelectric effect did not seem to agree with the wave description of light.
The "photoelectrons" emitted as a result of the photoelectric effect have a certain kinetic energy, which can be measured. This kinetic energy (for each photoelectron) is independent of the intensity of the light,[18] but depends linearly on the frequency;[20] and if the frequency is too low (corresponding to a photon energy that is less than the work function of the material), no photoelectrons are emitted at all, unless a plurality of photons, whose energetic sum is greater than the energy of the photoelectrons, acts virtually simultaneously (multiphoton effect).[23] Assuming the frequency is high enough to cause the photoelectric effect, a rise in intensity of the light source causes more photoelectrons to be emitted with the same kinetic energy, rather than the same number of photoelectrons to be emitted with higher kinetic energy.[18]
Einstein's explanation for these observations was that light itself is quantized; that the energy of light is not transferred continuously as in a classical wave, but only in small "packets" or quanta. The size of these "packets" of energy, which would later be named photons, was to be the same as Planck's "energy element", giving the modern version of the Planck–Einstein relation:
Einstein's postulate was later proven experimentally: the constant of proportionality between the frequency of incident light and the kinetic energy of photoelectrons was shown to be equal to the Planck constant .[20]
Atomic structure

In 1912 John William Nicholson developed[24] an atomic model and found the angular momentum of the electrons in the model were related by h/2π.[25][26] Nicholson's nuclear quantum atomic model influenced the development of Niels Bohr 's atomic model[27][28][26] and Bohr quoted him in his 1913 paper of the Bohr model of the atom.[29] Bohr's model went beyond Planck's abstract harmonic oscillator concept: an electron in a Bohr atom could only have certain defined energies
where is the speed of light in vacuum, is an experimentally determined constant (the Rydberg constant) and . This approach also allowed Bohr to account for the Rydberg formula, an empirical description of the atomic spectrum of hydrogen, and to account for the value of the Rydberg constant in terms of other fundamental constants. In discussing angular momentum of the electrons in his model Bohr introduced the quantity , now known as the reduced Planck constant as the quantum of angular momentum.[29]
Uncertainty principle
The Planck constant also occurs in statements of Werner Heisenberg's uncertainty principle. Given numerous particles prepared in the same state, the uncertainty in their position, , and the uncertainty in their momentum,
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk





















