
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
| Preferred IUPAC name
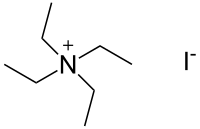
N,N,N-Triethylethanaminium iodide | |
| Other names
Tetamon iodide; Tetramon J; TEAI
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.000.615 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H20IN | |
| Molar mass | 257.159 g·mol−1 |
| Appearance | Colorless or yellowish crystalline solid |
| Density | 1.566 g/cm3[1] |
| Melting point | 280 °C (536 °F; 553 K) (decomposes) |
| soluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Tetraethylammonium iodide is a quaternary ammonium compound with the chemical formula C8H20N+I−. It has been used as the source of tetraethylammonium ions in pharmacological and physiological studies, but is also used in organic chemical synthesis.
Chemistry
Preparation
Tetraethylammonium iodide is commercially available, but can be prepared by the reaction between triethylamine and ethyl iodide.[2]
Structure
The crystal structure of tetraethylammonium iodide has been determined.[3] The crystal structure is a distorted wurtzite lattice. At the nitrogen atom, the coordination is a flattened tetrahedron. The N−C−C angle is slightly larger than the tetrahedral angle.
Synthetic applications
Examples include:
- Stereoselective formation of (Z)-diiodoalkenes by treatment of alkynes with ICl in the presence of tetraethylammonium iodide.[4]
- 2-Hydroxyethylation (attachment of −CH2−CH2−OH) by ethylene carbonate of carboxylic acids and certain heterocycles bearing an acidic N-H. For example, benzoic acid is converted to the ester, 2-hydroxyethyl benzoate, by treatment with ethylene carbonate in the presence of tetraethylammonium iodide.[5]
- Phase-transfer catalyst in geminal di-alkylation of fluorene, N,N-dialkylation of aniline and N-alkylation of carbazole using aqueous sodium hydroxide and alkyl halides.[6]
Toxicity
LD50: 35 mg/kg (mouse, i.p.); 56 mg/kg (mouse, i.v.)[citation needed]
See also
References
- ^ The Merck Index, 10th Ed., p.1316, Rahway: Merck & Co.
- ^ A. A. Vernon and J. L. Sheard (1948). "The solubility of tetraethylammonium iodide in benzene-ethylene dichloride mixtures." J. Am. Chem. Soc. 70 2035-2036. https://doi.org/10.1021/ja01186a015
- ^ E. Wait and H. M. Powell (1958). "The crystal and molecular structure of tetraethylammonium iodide." J. Chem. Soc. 1872-1875.
- ^ N. Hénaff and A. Whiting (2000). "Stereoselective formation of 1,2-diiodoalkenes and their application in the stereoselective synthesis of highly functionalised alkenes via Suzuki and Stille coupling reactions." J. Chem. Soc., Perkin 1 395-400.
- ^ T.Yoshino et al. (1977). "Synthetic studies with carbonates. Part 6. Syntheses of 2-hydroxyethyl derivatives by reactions of ethylene carbonate with carboxylic acids or heterocycles in the presence of tetraethylammonium halides or under autocatalytic conditions." J. Chem. Soc., Perkin 1 1266-1272.
- ^ G. Saikia and P. K. Iyer (2010)."Facile C-H alkylation in water: enabling defect-free materials for optoelectronic devices." J. Org. Chem. 75 2714-2717.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
