
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |

| |
| Names | |
|---|---|
| IUPAC name
Sodium hexanitritocobaltate(III)
| |
| Other names
Sodium cobaltinitrite
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.692 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
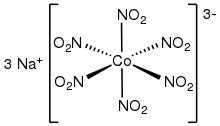
| Na3[Co(NO2)6 | |
| Molar mass | 403.933 g·mol−1 |
| Appearance | Yellow crystals |
| Density | 2.565 g/cm3 |
| Hazards | |
| GHS labelling:[1] | |
  
| |
| Danger | |
| H272, H315, H317, H319, H334, H335, H351 | |
| Safety data sheet (SDS) | JT Baker MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Sodium hexanitritocobaltate(III) is inorganic compound with the formula Na3[Co(NO2)6. The anion of this yellow-coloured salt consists of the transition metal nitrite complex [Co(NO2)63−. It was a reagent for the qualitative test for potassium and ammonium ions.[2]
Synthesis and reactions
The compound is prepared by oxidation of cobalt(II) salts in the presence of sodium nitrite:[3]
- 4 [Co(H2O)6(NO3)2 + O2 + 24 NaNO2 → 4 Na3[Co(NO2)6 + 8 NaNO3 + 4 NaOH + 22 H2O
Application for analysis of potassium
Although the sodium cobaltinitrite is soluble in water, it forms the basis of a quantitative determination of potassium, thallium, and ammonium ions. Under the recommended reaction conditions the insoluble double salt, K2Na[Co(NO2)6·H2O is precipitated and weighed.[4][page needed] In geochemical analysis, sodium cobaltinitrite is used to distinguish alkali feldspars from plagioclase feldspars in thin section.[5]

See also
References
- ^ "C&L Inventory". echa.europa.eu.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Glemser, O. (1963). "Sodium Hexanitritocobaltate(III)". In Brauer, G. (ed.). Handbook of Preparative Inorganic Chemistry. Vol. 1 (2nd ed.). New York, NY: Academic Press. p. 1541.
- ^ Vogel, A. I. (1951). Quantitative Inorganic Analysis (2nd ed.). Longmans Green and Co.
- ^ Bailey, E. H.; Stevens, R. E. (1960). "Selective staining of K-feldspar and plagioclase on rock slabs and thin sections". American Mineralogist. 45: 1020–1025. ISSN 1945-3027.
- ^ Brian N. Figgis; Alexandre N. Soboleva (2001). "Na3Co(NO2)6 at 293 and 10 K". Acta Crystallographica Section C. 57 (Pt 8): 885–886. doi:10.1107/S0108270101007995. PMID 11498599.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk


