
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
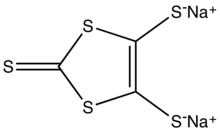
| Preferred IUPAC name
Disodium 2-sulfanylidene-2H-1,3-dithiole-4,5-bis(thiolate) | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C3Na2S5 | |
| Molar mass | 242.31 g·mol−1 |
| Appearance | yellow solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Sodium 1,3-dithiole-2-thione-4,5-dithiolate is the organosulfur compound with the formula Na2C3S5, abbreviated Na2dmit. It is the sodium salt of the conjugate base of the 4,5-bis(sulfanyl)-1,3-dithiole-2-thione. The salt is a precursor to dithiolene complexes and tetrathiafulvalenes.[1]
Reduction of carbon disulfide with sodium affords sodium 1,3-dithiole-2-thione-4,5-dithiolate together with sodium trithiocarbonate:
- 4 Na + 4 CS2 → Na2C3S5 + Na2CS3
Before the characterization of dmit2-, reduction of CS2 was thought to give tetrathiooxalate (Na2C2S4).[2]
The dianion C3S52- is purified as the tetraethylammonium salt of the zincate complex 2-. This salt converts to the bis(thioester) upon treatment with benzoyl chloride:[3][1]
- 2 + 4 C6H5COCl → 2 C3S3(SC(O)C6H5)2 + 2
Cleavage of the thioester with sodium methoxide gives sodium 1,3-dithiole-2-thione-4,5-dithiolate:
- C3S3(SC(O)C6H5)2 + 2 NaOCH3 → Na2C3S5 + 2 C6H5CO2Me
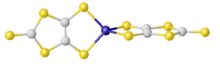
Na2dmit undergoes S-alkylation.[5] Heating solutions of Na2dmit gives the isomeric 1,2-dithioledithiolate.
References
- ^ a b "4,5-Dibenzoyl-1,3-dithiole-1-thione". Org. Synth. 73: 270. 1996. doi:10.15227/orgsyn.073.0270.
- ^ Dietzsch, W.; Strauch, P.; Hoyer, E. (1992). "Thio-oxalates: Their Ligand Properties and Coordination Chemistry". Coord. Chem. Rev. 121: 43–130. doi:10.1016/0010-8545(92)80065-Y.
- ^ G. S. Girolami, T. B. Rauchfuss and R. J. Angelici (1999) Synthesis and Technique in Inorganic Chemistry, University Science Books: Mill Valley, CA.ISBN 0-935702-48-2
- ^ W.T.A. Harrison; R.A. Howie; J.L. Wardell; S.M.S.V. Wardell; N.M. Comerlato; L.A.S. Costa; A.C. Silvino; A.I. de Oliveira; R.M. Silva (2000). "Crystal structures of three 2− salts: 2 (Q = 1,4-Me2-pyridinium or NEt4) and 2·DMSO. Comparison of the dianion packing arrangements in 2". Polyhedron. 19 (7): 821–827. doi:10.1016/S0277-5387(00)00322-3.
- ^ Niels Svenstrup; Jan Becher (1995). "The Organic Chemistry of 1,3-Dithiole-2-thione-4,5-dithiolate (DMIT)". Synthesis. 1995 (3): 215–235. doi:10.1055/s-1995-3910. S2CID 196762382.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
