
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
| IUPAC name
tetraiodoplatinum
| |
| Other names
Platinum tetraiodide, platinic iodide, platinum(4+) tetraiodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ECHA InfoCard | 100.029.280 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| I4Pt | |
| Molar mass | 702.702 g·mol−1 |
| Appearance | brown crystals |
| Density | 6.06 g/cm3 |
| Melting point | 130 °C (266 °F; 403 K) |
| decomposes in water | |
| Related compounds | |
Related compounds
|
Iridium tetraiodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Platinum(IV) iodide is a inorganic compound with the formula PtI4.[1] it is a dark brown diamagnetic solid and is one of several binary iodides of platinum.
Preparation
Platinum(IV) iodide can be prepared from the effect of iodine on platinum:[2]
- Pt + 2I2 → PtI4
It can also be obtained from the decomposition of hydrogen hexaiodoplatinate(IV) at 80 °C:
- H2[PtI6 → PtI4 + 2HI
Physical properties
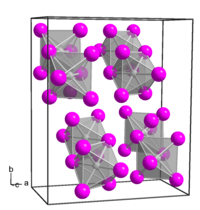
Platinum(IV) iodide forms dark brown crystals of several modifications:[3]
- α-PtI4, rhombic crystal system, spatial group P bca,[4] cell parameters a = 1.290 nm, b = 1.564 nm, c = 0.690 nm, Z = 8;
- β-PtI4, cubic crystal system, spatial group P m3m, cell parameters a = 0.56 nm, Z = 1;
- γ-PtI4, tetragonal crystal system, spatial group I 41/a, cell parameters a = 0.677 nm, c = 3.110 nm, Z = 8.
Platinum(IV) iodide decomposes in water. It is also soluble in ethanol, acetone, alkali, HI, KI, liquid NH3.[5]
Chemical properties
It decomposes when heated:
- PtI4 → Pt + 2I2
When dissolved in hydroiodic acid, platinum(IV) iodide forms hydrogen hexaiodoplatinate(IV):
- PtI4 + 2HI → H2[PtI6
References
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Wicks, Charles E.; Block, Frank E. (1963). Thermodynamic Properties of 65 Elements: Their Oxides, Halides, Carbides and Nitrides. U.S. Government Printing Office. p. 92. Retrieved 28 March 2024.
- ^ Macintyre, Jane E. (23 July 1992). Dictionary of Inorganic Compounds. CRC Press. p. 3510. ISBN 978-0-412-30120-9. Retrieved 28 March 2024.
- ^ Donnay, Joseph Désiré Hubert (1978). Crystal Data: Inorganic compounds 1967-1969. National Bureau of Standards. p. 153. Retrieved 28 March 2024.
- ^ "Platinum(IV) iodide, 99.95% (Metals basis), Pt 27.3% min., Thermo Scientific Chemicals, Premion | Fisher Scientific". Fisher Scientific. Retrieved 28 March 2024.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
