
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
| Other names
Iridium triiodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| I3Ir | |
| Molar mass | 572.930 g·mol−1 |
| Appearance | dark brown solid[1] |
| Density | 7.4 g·cm−3[1] |
| Solubility | insoluble in water and benzene[1] |
| Related compounds | |
Other anions
|
iridium(III) hydroxide iridium(III) chloride Iridium(III) bromide |
Other cations
|
Rhodium(III) iodide |
Related compounds
|
Iridium(IV) iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Iridium(III) iodide is an iodide of iridium, with the chemical formula of IrI3.
Preparation
Iridium(III) iodide can be obtained by reducing iridium(IV) iodide with hydrogen at 210 °C.[2] It can also be formed by the reaction of iridium dioxide[3] or iridium(III) hydroxide with hydrogen iodide.[4]
Chemical properties
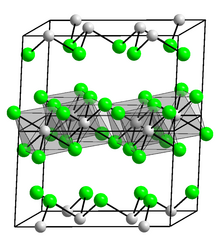
Iridium(III) iodide is a dark brown solid that is insoluble in water.[1] It is monoclinic like chromium trichloride.[5][6] Its trihydrate is yellow and can be dehydrated into the dihydrate or anhydrous form on heating. Iridium(III) iodide also has a monohydrate.[3][7]
References
- ^ a b c d Dale L. Perry (2016). Handbook of Inorganic Compounds, Second Edition. CRC Press. p. 523. ISBN 978-1-4398-1462-8.
- ^ Malatesia, Lamberto (March 31, 1961). PART I - STUDIES ON RHENIUM COORDINATION COMPOUNDS: HEXAISOCYANIDERHENIUM(I) SALTS; PART il - STUDIES ON IRIDIUM COMPOUNDS: ISOCYANIDE DERIVATIVES OF IRIDIUM, CARBONYL DERIVATIVES OF IRIDIUM IODIDES (PDF) (Report). Archived from the original (PDF) on 2018-10-30.
- ^ a b William Pettit Griffith (1967). The chemistry of the rarer platinum metals (Os, Ru, Ir, and Rh). Interscience Publishers. p. 241.
- ^ Raymond Eller Kirk; Donald Frederick Othmer; Herman Francis Mark (1963–1970). WHICH VOLUME?. Encyclopedia of Chemical Technology: Editorial board: Herman F. Mark, chairman, John J. McKetta, Jr. Donald F. Othmer. Interscience Publishers. p. 871. OCLC 2519460.[full citation needed]
- ^ R. Blachnik (2013). Taschenbuch für Chemiker und Physiker Band 3: Elemente, anorganische Verbindungen und Materialien, Minerale. Springer-Verlag. p. 494. ISBN 978-3-642-58842-6.
- ^ Brodersen, K. (February 1968). "Structure of β-RuCl3, RuI3, IrBr3, and IrI3". Angewandte Chemie International Edition in English. 7 (2): 148. doi:10.1002/anie.196801481. ISSN 0570-0833.
- ^ H. J. Kandiner (2013). Iridium. Springer-Verlag. ISBN 978-3-662-12128-3.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
