
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
| IUPAC name
iodoindium
| |
| Other names
Indium monoiodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.034.301 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| IIn | |
| Molar mass | 241.722 g·mol−1 |
| Appearance | red-brown solid |
| Density | 5.32 g/cm3 |
| Melting point | 365 °C (689 °F; 638 K) |
| insoluble | |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H302, H315, H319, H334, H335 | |
| P301, P302, P305, P312, P330, P338, P351, P352 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Indium monoiodide is a binary inorganic compound of indium metal and iodine with the chemical formula InI.[1][2]
Preparation
Indium(I) iodide can be obtained by reacting indium with iodine or indium(III) iodide in vacuum at 300 °C to 400 °C or with mercury(II) iodide at 350 °C.[3]
- 2In + I2 → 2InI[4]
- 2In + InI3 → 3InI
- 2In + HgI2 → 2InI + Hg
Physical properties
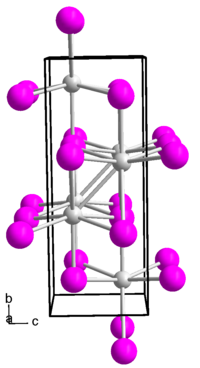
Indium(I) iodide forms a brown-red diamagnetic solid. Its melt is black. The compound has an orthorhombic crystal structure in the space group Cmcm (space group no. 63) with the lattice parameters a = 475 pm, b = 1276 pm, c = 491 pm.[5]
Chemical properties
Decomposes slowly with hot water:
- 2InI + H2O → InOH + HI
Reacts with water in the presence of oxygen:[6]
- 2InI + O + 3H2O → 2In(OH)2 + 2HI
References
- ^ "Indium(I) Iodide". American Elements. Retrieved 29 March 2024.
- ^ "Indium(I) iodide". Sigma Aldrich. Retrieved 29 March 2024.
- ^ Gasanov, A. A.; Lobachev, E. A.; Kuznetsov, S. V.; Fedorov, P. P. (1 November 2015). "Indium monoiodide: Preparation and deep purification". Russian Journal of Inorganic Chemistry. 60 (11): 1333–1336. doi:10.1134/S0036023615110066. ISSN 1531-8613. Retrieved 29 March 2024.
- ^ Rieke, Reuben D. (30 November 2016). Chemical Synthesis Using Highly Reactive Metals. John Wiley & Sons. p. 242. ISBN 978-1-118-92914-8. Retrieved 29 March 2024.
- ^ Fedorov, P P; Popov, A I; Simoneaux, R L (31 March 2017). "Indium iodides". Russian Chemical Reviews. 86 (3): 240–268. Bibcode:2017RuCRv..86..240F. doi:10.1070/RCR4609.
- ^ Satya, Prakash (2013). Advanced Chemistry of Rare Elements. S. Chand Publishing. p. 244. ISBN 978-81-219-4254-6. Retrieved 29 March 2024.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
