
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |
| Names | |
|---|---|
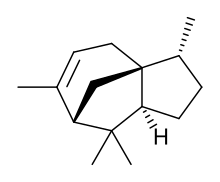
| IUPAC name
Cedr-8-ene
| |
| Systematic IUPAC name
(3R,3aS,7S,8aS)-3,6,8,8-Tetramethyl-2,3,4,7,8,8a-hexahydro-1H-3a,7-methanoazulene | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| 2207578 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.031.131 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C15H24 | |
| Molar mass | 204.357 g·mol−1 |
| Density | 0.932 g/mL at 20 °C[1] |
| Boiling point | 261–262 °C[1] |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H226, H304, H410 | |
| P210, P233, P240, P241, P242, P243, P273, P280, P301+P310, P303+P361+P353, P331, P370+P378, P391, P403+P235, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
| |
| Names | |
|---|---|
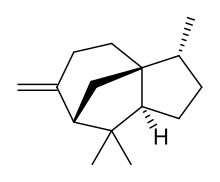
| IUPAC name
Cedr-8(15)-ene
| |
| Systematic IUPAC name
(3R,3aS,7S,8aS)-3,6,8,8-Tetramethyl-6-methylideneoctahydro-1H-3a,7-methanoazulene | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.031.131 |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C15H24 | |
| Molar mass | 204.357 g·mol−1 |
| Density | 0.932 g/mL at 20 °C[2] |
| Boiling point | 263–264 °C[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
Cedrene is a sesquiterpene found in the essential oil of cedar. The two isomers present in the oil are (−)-α-cedrene[3][4] and (+)-β-cedrene,[5] which differ in the position of a double bond.
See also
- Cedrol, another component of cedar oil
References
- ^ a b Sigma-Aldrich Co., (−)-α-Cedrene. Retrieved on 8 July 2012.
- ^ a b Sigma-Aldrich Co., (+)-β-Cedrene. Retrieved on 8 July 2012.
- ^ Lee, H. Y.; Lee, S.; Kim, D.; Kim, B. K.; Bahn, J. S.; Kim, S. (1998). "Total synthesis of α-cedrene: A new strategy utilizing N-aziridinylimine radical chemistry". Tetrahedron Letters. 39 (42): 7713–7716. doi:10.1016/S0040-4039(98)01680-3.
- ^ Takigawa, H.; Kubota, H.; Sonohara, H.; Okuda, M.; Tanaka, S.; Fujikura, Y.; Ito, S. (1993). "Novel allylic oxidation of α-cedrene to sec-cedrenol by a Rhodococcus strain". Applied and Environmental Microbiology. 59 (5): 1336–1341. Bibcode:1993ApEnM..59.1336T. doi:10.1128/aem.59.5.1336-1341.1993. PMC 182086. PMID 16348930.
- ^ Kerr, W. J.; McLaughlin, M.; Morrison, A. J.; Pauson, P. L. (2001). "Formal total synthesis of (±)-α- and β-cedrene by preparation of cedrone. Construction of the tricyclic carbon skeleton by the use of a highly efficient intramolecular Khand annulation". Organic Letters. 3 (19): 2945–2948. doi:10.1021/ol016054a. PMID 11554814.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
