
A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
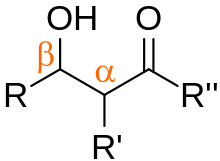
In organic chemistry, an aldol is a structure consisting of a hydroxy group (-OH) two carbons away from either an aldehyde or a ketone. The name combines the suffix 'ol' from the alcohol and the prefix depending on the carbonyl group, either 'ald' for an aldehyde, or 'ket' for a ketone, in which case it referred to as a 'ketol'. An aldol may also use the term β-hydroxy aldehyde (or β-hydroxy ketone for a ketol). The term "aldol" may refer to 3-hydroxybutanal.[1][2]
Aldols are the product of a carbon-carbon bond-formation reaction, giving them wide applicability as a pre-cursor for a variety of other compounds.
Synthesis and reactions

Aldols are usually synthesized from an aldol addition reaction using two aldehydes or an aldehyde and a ketone for a ketol.[1] These reactions may also be done intramolecularly to form 5 or 6 member rings or for stereoselective syntheses in the active area of asymmetric synthesis.
Aldols may also undergo a condensation reaction in which the hydroxy group is replaced by a pi bond. The final structure is a reactive α,β-unsaturated carbonyl compound, which can also used in a variety of other reactions:
- RC(O)CH2CH(OH)R' → RC(O)CH=CHR' + H2O
Applications
Aldols synthesized from two aldehydes are usually unstable, often producing secondary compounds such as diols, unsaturated aldehydes, or alcohols.[1] Hydroxypivaldehyde is a rare example of a distillable aldol.[3] The aldol 3-hydroxybutanal is a precursor to quinaldine, which is a precursor to the dye quinoline Yellow SS.[1]
Aldols are also used as intermediates in the synthesis of polyketide natural products and drugs such as Oseltamivir and Epothilone.[4][5][6][7]
See also
References
- ^ a b c d Kohlpainter, Christian; Schulte, Markus; Falbe, Jürgen; Lappe, Peter; Weber, Jürgen; Frey, Guido D. (15 January 2013). "Aldehydes, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry (7). doi:10.1002/14356007.a01_321.pub3. ISBN 9783527303854. Retrieved 1 April 2023 – via Wiley Online Library.
- ^ PubChem. "CID 21282929". National Center for Biotechnology Information. Retrieved 2023-04-15.
- ^ Zhang, Yanping; Mu, Hongliang; Pan, Li; Wang, Xuling; Li, Yuesheng (21 May 2018). "Robust Bulky Neutral Nickel Catalysts for Copolymerization of Ethylene with Polar Vinyl Monomers". ACS Catal. 8 (7): 5963–5976. doi:10.1021/acscatal.8b01088 – via ACS Publications.
- ^ Schetter, Bernd; Mahrwald, Rainer (2006). "Modern aldol methods for the total synthesis of polyketides". Angewandte Chemie International Edition. 45 (45): 7506–25. doi:10.1002/anie.200602780. PMID 17103481.
- ^ Ghosh, Arun K.; Dawson, Zachary L. (2009). "Synthesis of Bioactive Natural Products by Asymmetric syn- and anti-Aldol Reactions". Synthesis. 2009 (17): 2992–3002. doi:10.1055/s-0029-1216941. PMC 6233898. PMID 30443084 – via Thieme.
- ^ Ko, Ji S.; Keum, Ji E.; Ko, Soo Y. (15 October 2010). "A synthesis of oseltamivir (Tamiflu) starting from D-mannitol". J Org Chem. 75 (20): 7006–9. doi:10.1021/jo101517g. PMID 20866058 – via National Library Of Medicine.
- ^ Schinzer, Dieter (2004-06-24), Mahrwald, Rainer (ed.), "The Aldol Reaction in Natural Product Synthesis: The Epothilone Story", Modern Aldol Reactions (1 ed.), Wiley, pp. 311–328, doi:10.1002/9783527619566.ch7, ISBN 978-3-527-30714-2, retrieved 2024-04-15
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk
