A | B | C | D | E | F | G | H | CH | I | J | K | L | M | N | O | P | Q | R | S | T | U | V | W | X | Y | Z | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
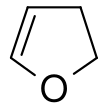
2,3-Dihydrofuran | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.013.407 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H6O | |||
| Molar mass | 70.091 g·mol−1 | ||
| Density | 0,927 g/mL | ||
| Boiling point | 54.6 °C (130.3 °F; 327.8 K)[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |||
2,3-Dihydrofuran is a heterocyclic compound with the formula C4H6O. It is isomeric with 2,5-dihydrofuran. 2,3-Dihydrofuran is one of the simplest enol ethers. It is a colorless volatile liquid.
Reactions
It undergoes lithiation upon treatment with butyl lithium. The resulting 2-lithio derivative is a versatile intermediate.[2][3]
Synthesis and occurrence
2,3-Dihydrofurans are intermediates in the Feist–Benary synthesis of furans from α-halogen ketones and β-dicarbonyl compounds.[4]
The 2,3-dihydrofuran ring can be synthesized by several methods. These routes usually involve cyclization or cycloaddition reactions of carbonyl compounds using metal-containing catalysts.[5][6] Iodine can also serve as a catalyst[7] as well as Raney nickel.[8]
References
- ^ Wilson, Christopher L. (December 1947). "Reactions of Furan Compounds. VII. Thermal Interconversion of 2,3-Dihydrofuran and Cyclopropane Aldehyde". Journal of the American Chemical Society. 69 (12): 3002–3004. doi:10.1021/ja01204a020.
- ^ Krzysztof Jarowicki; Philip J. Kocienski; Liu Qun (2002). "1,2-Metallate Rearrangement: (Z)-4-(2-Propenyl)-3-octen-1-ol". Org. Synth. 79: 11. doi:10.15227/orgsyn.079.0011.
- ^ Tschantz, M. A.; Burgess, L. E.; Meyers, A. I. (1996). "4-Ketoundecanoic Acid". Organic Syntheses. 73: 215. doi:10.15227/orgsyn.073.0215.
- ^ Gilchrist, Thomas L. (1997). Heterocyclic Chemistry (3rd ed.). Liverpool: Longman. pp. 209–212.
- ^ Kim N. Tu; Chao Gao; Suzanne A. Blum (2018). "An Oxyboration Route to a Single Regioisomer of Borylated Dihydrofurans and Isochromenes". J. Org. Chem. 83: 11204. doi:10.1021/acs.joc.8b01790.
- ^ Hao Liu; Zhenliang Sun; Kai Xu; Yan Zheng; Delong Liu; Wanbin Zhang (2020). "Pd-Catalyzed Asymmetric Allylic Substitution Cascade of But-2-ene-1,4-diyl Dimethyl Dicarbonate for the Synthesis of Chiral 2,3-Dihydrofurans". Org. Lett. 22: 4680. doi:10.1021/acs.orglett.0c01483.
- ^ Domenic P. Pace; Raphaël Robidas; Uyen P. N. Tran; Claude Y. Legault; Thanh Vinh Nguyen (2021). "Iodine-Catalyzed Synthesis of Substituted Furans and Pyrans: Reaction Scope and Mechanistic Insights". J. Org. Chem. 86: 8154. doi:10.1021/acs.joc.1c00608.
- ^ Evgeny V. Pospelov; Alexander V. Zhirov; Baglan Kamidolla; Alexey Yu. Sukhorukov (2023). "Reductive Denitrogenation of Six-membered Cyclic Nitronates to Densely Substituted Dihydrofurans with Raney® Nickel/AcOH System". Adv. Synth. Catal. 365: 8154. doi:10.1002/adsc.202300573.
Text je dostupný za podmienok Creative Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších podmienok. Podrobnejšie informácie nájdete na stránke Podmienky použitia.
Antropológia
Aplikované vedy
Bibliometria
Dejiny vedy
Encyklopédie
Filozofia vedy
Forenzné vedy
Humanitné vedy
Knižničná veda
Kryogenika
Kryptológia
Kulturológia
Literárna veda
Medzidisciplinárne oblasti
Metódy kvantitatívnej analýzy
Metavedy
Metodika
Text je dostupný za podmienok Creative
Commons Attribution/Share-Alike License 3.0 Unported; prípadne za ďalších
podmienok.
Podrobnejšie informácie nájdete na stránke Podmienky
použitia.
www.astronomia.sk | www.biologia.sk | www.botanika.sk | www.dejiny.sk | www.economy.sk | www.elektrotechnika.sk | www.estetika.sk | www.farmakologia.sk | www.filozofia.sk | Fyzika | www.futurologia.sk | www.genetika.sk | www.chemia.sk | www.lingvistika.sk | www.politologia.sk | www.psychologia.sk | www.sexuologia.sk | www.sociologia.sk | www.veda.sk I www.zoologia.sk